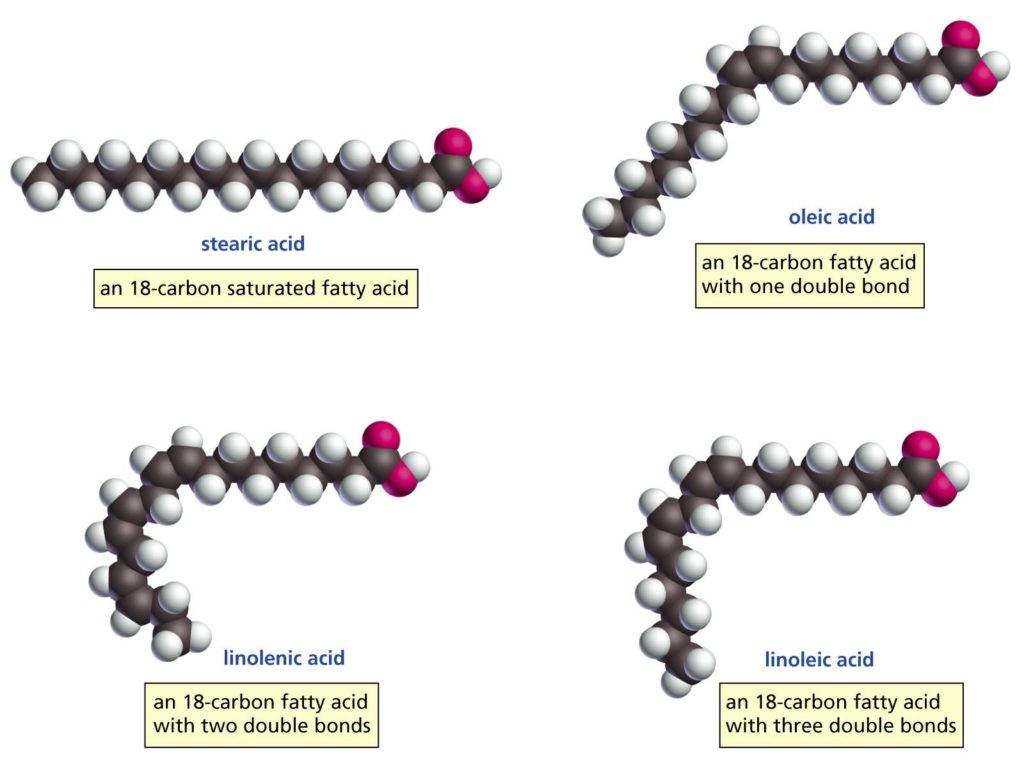
There are three basic kinds of fat, Saturated Fat (SFA), Monounsaturated Fat (MUFA) and Polyunsaturated fat (PUFA). Fats are also called “fatty acids”, hence the term PUFA: PolyUnsaturated Fatty Acid. Fats are a chain of carbon atoms with hydrogen atoms bonded to them. Each carbon in a chain can bond to two hydrogen atoms. This gives it four bonds total – two hydrogen bonds plus a bond to the carbon on either side of it. In a “saturated” fat, the carbons are saturated with hydrogen, meaning that every carbon is bonded to two hydrogens. Saturated fats are straight and they pack together tightly, which gives them a high melting point. The melting point of pure stearic acid is over 150 degrees Fahrenheit! Saturated fat is basically shaped like a long, straight skinny hot dog. If hot dogs are all lined up they pack together well.
In an “unsaturated” fat, two neighboring carbons are each only bonded to a single hydrogen and they share a double bond with each other. This still leaves the carbon reasonably happy with four bonds total – one bond to the carbon on the right, two bonds to the carbon on the left and one with hydrogen. This double bond gives the molecule a “kink”. A monounsaturated fat (MUFA) has a single double bond between two of the carbons and a shape like a boomerang. Boomerangs don’t pack together as well as hot dogs and so monounsaturated fats slide around each other and are more liquid. This is why olive oil, which is composed of 70% MUFA, is a liquid at room temperature and butter, mostly SFA, is a solid.
“Polyunsaturated” fats have multiple double bonds. The multiple kinks give them almost a C shape. C’s don’t pack at all well together and polyunsaturated fats have a lower melting point yet.
Chain Length Matters
The other way that fats vary is in the number of carbons in the chain.
The vast majority of fats that are consumed in humans – SFA, MUFA and PUFA – are 16 or 18 carbons long. Even numbered carbon chains are far more common although odd numbered chains exist. There are some fats that are longer than 18 carbons, these are mostly PUFA, such as the Omega 3 DHA found in fish oil.
Interestingly, humans have elongase and desaturase enzymes that can make PUFA longer and less saturated, but SFA and MUFA consumed by humans stays as SFA or MUFA. SFA can be converted to MUFA but not the other way around.
A unique thing about SFA is that there are food sources that contain significant amounts of saturated fats with carbon lengths shorter than 18 carbons, all the way down to the 4 carbon butyrate, found mainly in butter. Longer hot dogs pack tighter together than cocktail weenies and so short chain saturated fats have much lower melting points than 16 or 18 length fats. MCT oil – an oil derived from coconut oil – contains only 8 and 10 length carbon chains and are liquids at room temperature. These short chain saturated fats are what make coconut oil soft at room temperature compared to 16 and 18 carbon length saturated fats which are very hard indeed!
Another common source of short chain saturated fats is butter. Short chain fats make up around 20% of the saturated fats in butter and butter is the only common source of the four carbon butyric acid. Goat’s milk has a higher fraction of saturated fats with 6 to 10 carbon chain lengths than cow’s milk, which is what gives goat cheese its unique flavor and why those fats are all named after goats. Butter is a unique fat in that it is actually 20% water. Butter is a water and fat emulsion, much like mayonnaise. The short chain fats and the fact that it’s an emulsion give butter its creamy texture at room temperature.
A Lurking Danger in PUFA
This section is about fat oxidation. For background you may want to start with A Very Gentle Introduction To Oxidation.
Saturated fats (SFA) and monounsaturated fats (MUFA) are both pretty happy from an energetic perspective and won’t easily surrender any of their electrons to oxygen. But polyunsaturated fats (PUFA) are different. A free radical induced process called autoxidation leads them to become lipid peroxides, dangerous free radicals that can do damage to surrounding proteins and DNA. This is one of the reasons vegetable oils are high in vitamin E, an antioxidant. Antioxidants are “electron donors” and they step in to quickly give an electron to any superoxide molecules that come sniffing around to try to snatch an electron away from PUFA, thus stopping the PUFA from becoming oxidized.
Nonetheless, the threat of PUFA becoming oxidized is always there and PUFA oxidation is undoubtedly a bad thing. Which is why animals don’t make PUFA.
Why Do We Need All of These Different Fats?
Different organisms all face different environmental challenges, and so having fats with different melting points is crucial. Proper membrane fluidity is crucial to all living things. In a warm blooded mammal, such as a human, the internal temperature is very tightly controlled. The means that our cellular membranes can be composed of a roughly 50:50 blend of saturated and monounsaturated fats and membrane fluidity will be maintained. Warm blooded animals don’t need much PUFA at all and so our bodies don’t make them. Why make something that’s a threat to oxidize if you don’t have to?
But if we were a steelhead trout migrating into a creek in November it’s a very different story! The body temperature of the trout is the same as the water she is in. If her fat was 50:50 saturated and monounsaturated she would freeze up like a brick! This is why salmon and trout – fish from cold Northern waters – are full of highly unsaturated Omega 3 fats. Of course fish don’t make the Omega 3’s, they get them from planktons, which face the same issue of maintaining membrane fluidity in a cold environment and so they manufacture the Omega 3 fat which bioaccumulate up the food chain to the trout.
The same thing is true of a corn seed. When seeds go into the ground in May (this is when it is planted in upstate New York where I’m writing this) into 55 degree soil, the corn seed needs to start growing and for this the corn seed needs to have fluid fat, which is why corn oil is comprised mostly of the doubly unsaturated linoleic acid – PUFA.
So why do trout have Omega 3 and corn seeds have omega 6 fats? The common Omega 3 fats have at least three double bonds, which makes them very fluid even at freezing temperatures but all of those double bonds make them extremely prone to oxidation, which is why flaxseed oil is sold in brown bottles (to prevent damaging light radiation) and corn oil is sold in clear bottles. The trout has to keep swimming even in 32 degree water. The corn seed has the luxury of waiting for the soil to hit 55 degrees to do its thing.
Tropical plants don’t have to deal with the cold environment of a trout or and corn seed and so they produce fat that is much more saturated. Again, why make much PUFA if you don’t have to? Since tropical plants have no mechanism for regulating their body temperature, they still have to maintain membrane fluidity at temperatures much lower than a warm blooded mammal would. For this reason palm oil is about 9% PUFA – as opposed to the fat of a cow which is 3-4% PUFA. Coconut oil has taken the rather unique approach of producing lots of short chain saturated fats for fluidity and so it is one of the lowest sources of PUFA in commonly eaten plant fats. Cocoa butter is very low in PUFA and high in long chain fats. It is therefore a very hard fat and cocoa seeds won’t germinate much below 80 degrees Fahrenheit (27 Celsius), whereas a coconut will germinate at 70 degrees Fahrenheit (21 Celsius).
Eat Like A Human
Humans, pigs and chickens can, in fact, only make saturated fat from dietary sources like starch, sugar, alcohol and protein. The process of making our own fat is called de novo lipogenesis. Once the saturated fat is made, the amount that remains saturated versus the amount that becomes monounsaturated is controlled by an enzyme called Stearoyl-CoA Desaturase-1 or simply SCD1.
The ONLY dietary component that can effect the fat ratios in our bodies are MUFA and PUFA. We are what we eat and we only get PUFA from our diet, so if we eat PUFA, the amount in our fat stores and membranes go up. The same can be said of MUFA, although if we eat MUFA, the body will produce less SCD1 to try to remain at the correct balance of SFA and MUFA.
If we eat a diet of saturated fat, starch, sugar, alcohol and protein our body fat will remain relatively saturated. Once we start consuming MUFA and PUFA we will become considerably less saturated.
All Fats Are Blends
All naturally occurring fats are blends of SFA, MUFA and PUFA., as you can see from the following table. In same cases the percentages don’t add to 100, I’m pulling the data directly from the USDA, so I’m not sure why that is.
| SFA | MUFA | PUFA | |
| Coconut Oil | 82% | 6% | 2% |
| Stearic Acid Enhanced Butter Oil | 77% | 20% | 3% |
| Butter | 68% | 28% | 4% |
| Cocoa Butter | 60% | 33% | 3% |
| Beef fat (Ribeye) | 46% | 50% | 4% |
| Palm Oil | 50% | 37% | 9% |
| Lard (From a pig I raised, wheat finished) | 45% | 48% | 7% |
| Lard | 41% | 47% | 12% |
| Lard (16% Dry Distiller’s Grains) | 31% | 41% | 28% |
| Chicken Fat | 31% | 45% | 24% |
| Olive Oil | 12% | 77% | 11% |
| Canola Oil | 7% | 63% | 28% |
| Corn Oil | 14% | 29% | 58% |
| Sunflower Oil | 11% | 20% | 69% |
Fat Nomenclature
Often fats will be abbreviated like 18:0. That is the symbol for stearic acid. It is 18 carbons long and contains 0 double bonds. Linoleic acid – Omega 6 PUFA – is 18:2 n-6, which is to say that it is 18 carbons long, it has two double bonds and the FIRST double bond is at the sixth carbon in the chain. Oleic acid from olive oil is 18:1 n-9 – 18 carbons with one double bond at the ninth carbon. Fats are often named after the most common natural source, so OLeic acid is from OLives.
| Fat | Named After | Abbreviation |
| Saturated Fats (SFA) | ||
| Stearic Acid | Beef Steer | 18:0 |
| Palmitic Acid | Palm Oil | 16:0 |
| Myristic Acid | Nutmeg | 14:0 |
| Lauric Acid | 12:0 | |
| Capric Acid | Goats, Gives the unique flavor in goat cheese | 10:0 |
| Caprylis | Goats | 8:0 |
| Caproic Acid | Goats | 6:0 |
| Butyric Acid | Butter | 4:0 |
| Monounsaturated Fats (MUFA) | ||
| Oleic Acid | Olives | 18:1 n-9 |
| Polyunsaturated Fat (PUFA) | ||
| Linoleic Acid | Linon (flax) | 18:2 n-6 |
| Linolenic acid | Linon (flax) | 18:3 n-3 |
| DHA | 22:6 n-3 |
Triglycerides
When fats are in storage in a fat cell, three of them are linked together by a molecule called glycerol using a bond called an ester bond. Lipases (enzymes) remove the fats from the glycerol before they can be metabolized. At this point, the NEFA (Non Esterified Fatty Acids) can move into the mitochondria for metabolism or be released into the blood as Free Fatty Acids (FFA). Of course, fats aren’t water soluble and can’t just float around in the blood. They are first attached to albumin, a large, water soluble protein to make them soluble. Hungry cells in the body can use these FFAs for fuel.

By far the best explanation of fatty acids I’ve seen.
There is an error in the image, though. The descriptions of the linolenic and the linoleic acids are switched. For linolenic acid, it says “an 18-carbon fatty acid with _two_ double bonds”, but it should say “…with _three_ double bonds”.
Additionally, the two bottom molecules should be switched around, so the number of bonds increases from left to right.
Haha! Thanks for the feedback. Unfortunately I borrowed the image and I’m not much of an artist.
Any data on the FA composition of bear fat, mutton tallow, or duck fat?
I have been reading through this blog for the last couple of days and am very intrigued by the ROS theory. Please continue.
I recently saw a paper suggesting that bear fat, a staple of native Americans, was VERY low in PUFA and high in saturated fat. More than I would have suspected from a monogastric omnivore so I’m skeptical. Mutton tallow is probably a good choice, especially if it’s from suet – kidney fat. Duck fat will be fairly low in saturated fats and the PUFA content depends on what it is fed.
Wouldn’t a bear eating lots of sugar (honey, fruits) have a lot of saturated fat?
Presumably? I have no idea what the overall composition of a bear diet looks like.
I live in bear country . They are omnivores , eating what is in season – acorns in the fall, small animals, grubs and larvae.
Really enjoying all of the fire in a bottle series! All of your posts have been extremely thought provoking! I’ve appreciated every one…Thank you!
Question for you: Since PUFAs are more prone to oxidation due to their double bonds, why do omega 3s generally seem to be anti-inflammatory for people ie. protective against cardiovascular disease and cancer while omega 6s are pro-inflammatory and are involved in pathogenesis of heart disease and cancer?
Related: If Omega 6: Omega 3 ratio is predictive for cardiovascular disease, do you think that similarly a SFA/PUFA ratio in diet would be important as well?
The omega 3/6 discussion is a pretty deep rabbit hole and I won’t go too far down it here. I think it’s too simplistic to say 6’s are pro-inflammatory and 3’s are anti. If you want to destroy the liver of a mouse, feed it fish oil and alcohol.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6062209/
That’s not to say that Omega 3 fats are bad, but understand that they are HIGHLY prone to oxidation and tread carefully. I’m sure I’ll have a lot to say on this later.
Does the butter have to be grass-fed or is regular supermarket butter fine?
Also, chicken and pork – I heard you say on Biohackers lab that the fat in these meats isn’t healthy because of what these animals are fed. Is organic chicken and pork better or do they just feed them organic “poison” (seed oils)?
Whether or not you use grassfed or conventional butter depends on your preference and your wallet. I like the grassfed.
Organic chicken is fed just as much soybean oil as conventional. Organic pigs are corn finished but at least avoid the disastrous DDGS.
Brad