I believe that obesity is a fuel partitioning disorder. I’ve argued that when you pack on fat, you essentially “lose” that fuel into your fat tissue. If you don’t have access to fuel, you become hungry and you eat more.
In my last article, I started to delve into the mechanism of this. One thing that is shared between obese humans and animals fattening for winter is elevated PDK4 in skeletal muscle. This blocks the ability of muscle tissue to utilize glucose. So blood glucose is high but you can’t USE it!
The paper1 from the last article has a gem of a chart illustrating this in practice.
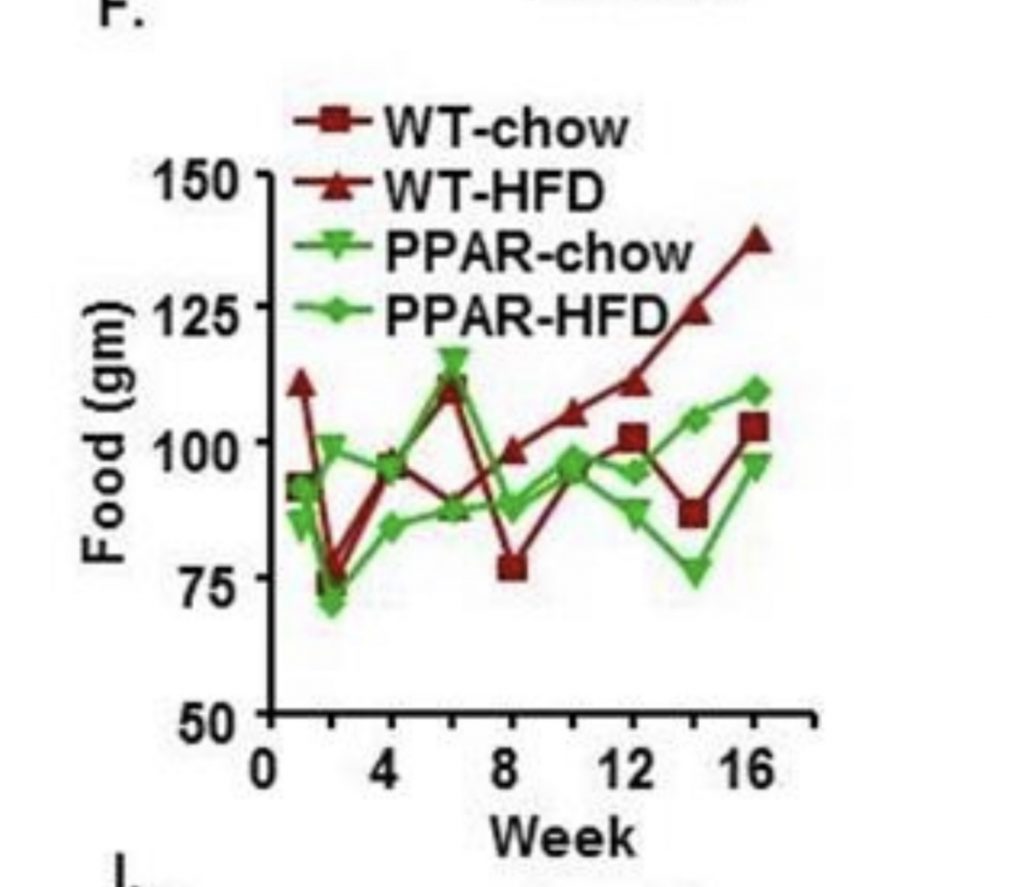
There are four groups of mice. The red squares are “normal” mice eating low fat diet. The red triangles are normal mice eating a diet with 60% of calories as high-PUFA lard with some soybean oil. The green triangles are mice who lack PPAR gamma – who therefore will not become torpid – eating a low fat diet. The green diamonds are the mice lacking PPAR gamma on the high fat diet.
And here are the weights of the mice. Remember again that these mice aren’t really normal, this is a special straim (C57BL/6) that is known to reliably become fat when fed a high fat diet.
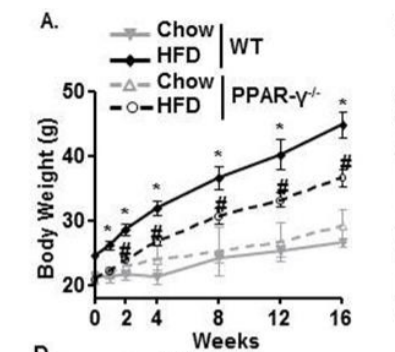
Here are the weights of the mice. The “normal” mice on the high fat diet is the solid black line and they are easily the fattest. The mice lacking PPAR gamma – dashed black line – also become fat, but not nearly so much. The ROS Theory Of Obesity would predict that an obesity prone organism on a mixed diet of starch and mostly unsaturated fat will gain weight while remaining glucose tolerant and this is what we see.
Let’s think about the two groups of mice on the high fat diet. For the first 6 weeks their food consumption is similar. From that point on the food consumption of both groups of mice rises steadily, but the food consumption of the torpid, insulin resistant mice with a functional PPAR gamma gene far surpasses the consumption of all other mice. They are overeating.
Why are they overeating? Is it because their food is too palatable and too low in fiber and they lack willpower? If that was the issue, the mice lacking PPAR gamma would overeat just as much. But they don’t.
Interestingly, their food consumption rises in parallel with their blood glucose – an indirect indicator of their degree of torpor. Torpid hibernating animals become hyperphagic in the fall -they overeat.
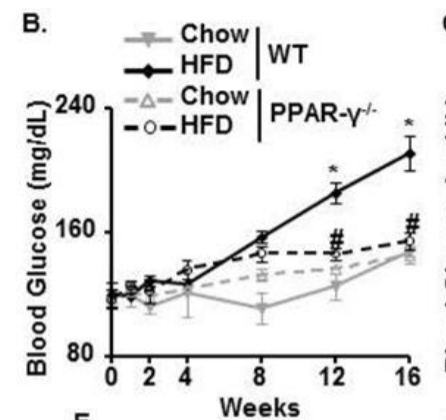
So as the skeletal muscles of the mice become torpid and lose access to glucose as a fuel the mice consume more food. Lipogenic genes are turned on in the liver and so the glucose is being turned into fat in the liver rather than burned as glucose in the muscle.
We could argue about the exact mechanism by which the appetite of the torpid mice is increased, but what is clear is this: the mice increased their caloric intake in response to the activation of PPAR gamma by oxidized PUFA. They are overeating because they have a torpid metabolism and not the other way around.
- 1.Sikder K, Shukla SK, Patel N, Singh H, Rafiq K. High Fat Diet Upregulates Fatty Acid Oxidation and Ketogenesis via Intervention of PPAR-γ. Cell Physiol Biochem. Published online 2018:1317-1331. doi:10.1159/000492091

Yep, we agree on both of those things. You’re not fat because you’re inactive you’re an active because you’re fat. You aren’t fat because you overeat, you overeat because you’re fat… actually you’re torpid Because you’re eating the wrong kinds of fat that activate the wrong genes.
Ultra-processed food is killing humanity. The latest studies have shown that. And now you’ve teased out the mechanism.
I can’t think of a diet that “works” for fat loss that doesn’t fit this paradigm. Can you?
How does all this truly translate to humans though?? I seem to be one of those few rare people that has eaten very very little processed food in my lifetime (stupid orthorexia and finally recovered from that thankfully) and yet I still got ‘fat’ after 40. Not extremely fat but definitely have unhealthy levels reflected in ongoing blood markers. Some of that is due to age and other factors outside my control (insomnia, 2 young kids and a husband all with untreated ADHD), but I still haven’t given up finding the key to stopping the upward stepping of body fat without having to suffer. Sorry didn’t mean to give my life fat story lol. But following a very low PUFA diet and adding cocoa butter in coffee, although admittedly very enjoyable, seems to have had the opposite effect on the scale to that desired. So I wonder how much this theory can really apply to humans. It does baffle me about the French (and many other people before the 80s) – not much obesity around. But it did exist….I wonder about those individulas too…and maybe I am one of them! I know you mentioned a study or two done on humans…are there any more? Or am I completely ignoring a whole stash of studies? I very much enjoy reading all this though….
So there are a whole host of factors that can influence the up-regulation of the lipogenic genes. I’m actually working on a series of posts about exactly this and what can be done about it. Stayed tuned. Here is very brief teaser: the aryl hydrocarbon receptor (AHr) is triggered by environmental toxins called dioxins or Persistent Organic Polutants. Polyyclic Aromatic Hydrocarbons from charred meats, cigarette smoke and car exhaust trigger another receptor called PXR or SXR. Getting rid of AHR prevents diet induced obesity. These “receptors” can be combatted by stimulating AMPK.
Brad
I see Brad had mentioned pollutants. I am over 50 and into menopause, and despite a lifelong commitment to unprocessed food, and avoiding seed oils and most grains for 10 years, I gained 20+ pounds in the past couple years. Some of it was menopause related (e.g. my thyroid dipped; I’m now on meds). But I tried counting calories, and despite having a watch that tracked my activity levels and recommended I eat between 1550 and 1700 calories per day to lose a pound a week, did not lose weight until I averaged 850 per day, and then I lost 1/2 pound a week. What on earth? Eating more – lots more – did not make me gain more weight either.
I’ve been trying the things on this blog for months, but I’ve only lost weight when I travel. My home has a damp crawlspace and sometimes I have to fight mold. I’m suspecting that mold is an issue in my persistent fat. It’s almost all abdominal. I just had a doctor’s appointment, and she agrees that mold could be the culprit.
Would have been impactful to see the results of a high SFA, low PUFA diet along with the rest on those graphs. For anyone wondering, they sort of did that here: https://fireinabottle.net/the-amount-of-polyunsaturated-fat-in-the-modern-american-diet-fattens-up-mice-without-additional-calories/ — so we know the effects, but just saying it would have been nice to see it again compared to the rest in this study.
I can’t help but look at the chow diet line for blood glucose and body weight and think devil’s advocate: “well, isn’t that an alternative?” That is, fixing a broken metabolism with a low fat diet? But the chow was only 13% kcal from fat, which seems impossibly low for western humans eating anything close to a familiar palatable diet in order to maintain compliance with that diet. Maybe it could be thought of as a proxy for starch eaters (e.g. Nanan, China), which we know through Brad’s other articles end up with saturated fat stores through de novo lipogenesis.
Yes, I actually did a short dietary trial with a Very Low Fat diet, just to see what would happen while using sterculia oil and citrus polyphenols. It actually worked OK, although I moved on to try something else. Food was pretty good: Cubed potatoes boiled in broth until it cooked down into a kind of gravy served with a lean Firebrand Meats low-PUFA pork rump steak. Or Noodles in a Ramen broth with spicy chili sauce.
Brad
This idea has been pushed by Gary Taubes for like nearly 20 years. One problem is – if obesity is a fat partitioning disorder that then why are most of the single nucleotide polymorphisms that have been identified to affect obesity in centrally acting genes in hunger pathways. You may like our contrast of the carbohydrate insulin model which is essentially the fat partitioning idea, and the energy balance model, published in Science a few months back.
Speakman, J.R. and Hall, K.D. (2021) Carbohydrates, insulin and obesity
Science 372: 577-578 DOI: 10.1126/science.aav0448
Yes, I have certainly been influenced by Taubes in my thinking on this, but this was such a nice example of it that I stumbled upon!
Thanks for the link, looks interesting but pity about the lack of public access! As to the polymorphisms being involved in central acting hunger pathways, I think this concept is oversold. SCD1, sirt3, polymorphisms correlate with obesity:
https://onlinelibrary.wiley.com/doi/full/10.1038/oby.2007.206
AMPK polymorphisms are associated with diabetes:
https://www.sciencedirect.com/science/article/abs/pii/S0306453011001909
SIRT3 polymorphisms are associated with metabolic syndrome:
https://www.cell.com/molecular-cell/fulltext/S1097-2765(11)00543-0?_returnURL=https%3A%2F%2Flinkinghub.elsevier.com%2Fretrieve%2Fpii%2FS1097276511005430%3Fshowall%3Dtrue
Those three examples are the first three I thought of. 3/3 were associated with obesity/diabetes and/or metabolic syndrome.
Brad
Link to their paper here:
https://www.researchgate.net/publication/351385114_Carbohydrates_insulin_and_obesity
Here is a long and interesting twitter thread on the obesity epidemic.
https://threadreaderapp.com/thread/1412827768329494528.html
Also, I have started TCD this week and already lost 2 inches off my belly fat
Wow, congrats!
So what’s the solution? The only thing I’ve been able to do in the last year, where I’ve actually seen a difference, is fasting. I did three consecutive five day, M-F fasts, which I broke on the weekends, over the prior three weeks. I lost a significant amount of weight. Felt pretty great, too. Taking a break this week. But will get right back to it next week.
The data presented here carefully unpack all the mechanisms and it’s very impressive, but what concerns me is that once you become overweight you seem to be stuck there and there’s no way to get your weight back down because of the hormonal an enzymatic imbalances that you create.
What is the path forward?
Keep an eye out the next few weeks. I’m going to present a few more problems first and then start talking about solutions…
Curious about something regarding fat gain and PPAR gamma.
How do the PPARg-knockout mice gain fat? Is it dietary fat (already triglycerides in chylomicrons or VLDL) being stored under influence of insulin?
Or put differently- The transcription factors I liken to calling up a menu of gene transcripts like ordering a #1 from McDonalds, vs a #2 or something; the signal is simple but the end result is a lot of different outputs. We know PPARg produces fat synthesis, desaturase and some other enzymes. What else? Among the cocktail of proteins/enzymes produced by PPARg activation your thesis so far reads: PPAR gamma is a “torpor, sloth & gluttony fat synthesis & gain” signal. Curious if PPARg has any other roles – would it ever be appropriate to see PPARg active in the springtime, as opposed to autumn/harvest time?
Hi Brad,
Interesting article. Trying to raise my own hogs and chicken for personal consumption. What diet would you recommend I feed them? Are there other sources of protein low in PUFA’s?
The easiest thing to find as a base diet for pigs that is low in PUFA is ground wheat. If you supplement with the correct amino acids: lysine, threonine, methionine, there is enough protein in whole wheat for pigs over ~150 lbs. Wheat has half the PUFA of corn.