Introduction
This is part of my Torpor, Sloth and Gluttony series where I will argue that obesity is caused not by sloth and gluttony but by specific metabolic processes.
Leptin is a hormone produced by fat cells. The fatter one gets, the more circulating leptin one has. Leptin is the master hormone whose job is meant to prevent obesity by decreasing food intake and increasing metabolic rate. This article digs into exactly HOW leptin affects metabolic rate.
It seems that leptin makes our skeletal muscle inefficient by increasing an uncoupling protein known as UCP3. This statement is controversial but I think the evidence is strong. By inefficient I mean that it takes more calories to do the same job. If you respond to leptin you will burn more calories when doing simple activities. You will become bad at walking, from an energy efficiency perspective.
Leptin Raises Your Metabolic Rate
I love scientific papers that tell a story and I love science that is repeated. The whole idea of science is repeatability, of course. This paper1 gives us both a narrative and repeatability. The same research group did a pilot study with four subjects published in 20022 and followed up with this study of 10 subjects in 2005. Here’s the narrative:
Subjects were kept as inpatients of a hospital research ward. They were put on a liquid diet and the number of calories given to each participant was raised or lowered until they reached weight stability. Subjects were kept at this weight for a number of weeks while leptin levels, thyroid hormone levels, total energy expenditure per 24 hours, resting metabolic rate and metabolic rate while riding a stationary bike at different levels of exertion were all measured. Then caloric intake of subjects was dropped to 800 calories per day until they had lost 10% of their body weight. Over several weeks the number of calories given to each subject was re-adjusted until weight stability at the lower weight was achieved. All of the tests were repeated. At this point the subjects caloric requirements (total energy expenditure) had dropped by an average of 22% – far in excess to the proportion of body weight lost – and leptin levels had dropped by 29%.
The final leg of this experiment was to inject the subjects with small amounts of leptin, just enough to bring circulating levels up to the pre-weight loss levels. When this was done, subjects began losing weight again while consuming the same number of calories. Calories were adjusted back upward to again achieve weight stability. The new caloric burn( total energy expenditure ) rebounded to within 5% of starting levels. Leptin restored the metabolic rate of the dieters.
This grueling protocol lasted on average 28 weeks in a hospital ward for the subjects!! Without a single bite of solid food!!! That is quite a sacrifice for science. You can see why they only used 10 volunteers. I would have made a daring escape from the hospital in the wee hours of the morning on like day 2. The good news is that the responses of the volunteers – 2 lean males, 1 lean female, 3 obese males and four obese females – were remarkably consistent and match the previous study. This is repeatable.
Anyone who has tried to lose weight with the idea of “just eat less” can see the cruelness of these findings. Losing 10% of your body weight causes a whopping 22% decrease in caloric needs! If you’re a 200 lb male with a target weigh of 160 and you lose 10%, you’ll now weigh 180 lbs (20 lbs lost). Now you have to lose another 20 lbs but your caloric burn rate has plummeted far in excess of the weight lost! The last 20 lbs will come off much slower and more painfully than the first 20. The first 20 lbs lost (in this study) took 8 weeks of an 800 calorie liquid diet. What will the next 20 take?!
Leptin Makes You Bad At Walking
The authors could have said something like “Leptin measurably increases metabolic rate in actual, live, normal human beings”, dropped the mic and walked away.
But they were interested in HOW leptin increases metabolic rate, and you should be as well. The opening line of the paper is “Maintenance of a reduced body weight is accompanied by decreased energy expenditure that is due largely to increased skeletal muscle work efficiency“.
A less confusing way to say this is that “leptin makes you burn more calories to do the same amount of work”. In particular, the subjects became very efficient at slowly pedaling a bike at a rate that would generate 10 Watts of electrcity.
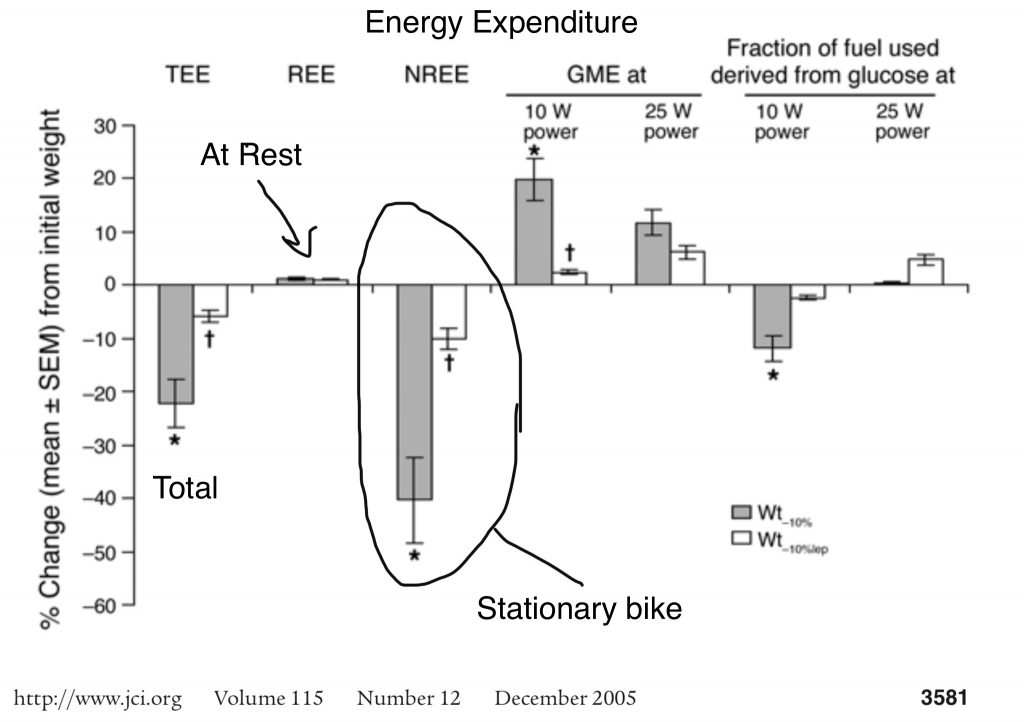
The gray bars are the change in energy expended after subjects lost 10% body weight. The white bars are energy expended after losing 10% body weight and then supplementing to replace leptin. As you can see, total energy expenditure dropped dramatically but mostly rebounded with leptin. Perhaps more surprising is that resting energy expenditure after weight loss didn’t change AT ALL. The difference in energy usage was due to the fact that subjects burned MUCH less calories while doing normal tasks like walking around or slowly riding a stationary bike. Leptin made them burn more calories while “doing stuff”.
So weight subjects were very EFFICIENT at riding a bike but very INEFFICIENT at burning calories. All of the action was taking place in muscle tissue.
UCP3 Is Down-Regulated With Weight Loss
What is the molecular target of leptin in muscle? How is it making movement less efficient?
Even before the above experiment was carried out, the same research group had shown that a 10% reduction in body weight resulted in a 28% drop in a protein called uncoupling protein 3 (UCP3).3 Could that be the target?
It had been known that a protein called uncoupling protein 1 (UCP1) allows mitochondrial uncoupling in the Brown Adipose Tissue (BAT) of rodents which allows the rodents to run their metabolism very inefficiently.4,5 UCP1 short circuits the mitochondrial “battery”, allowing protons to run back down through into the mitochondria, releasing heat without doing any work. Fat and carbohydrates are burned, but no ATP is formed. This is a cool trick in mice, but humans have very little brown adipose tissue.
All signs point to the target being UCP3. UCP3 is in the family of uncoupling proteins, which are known to create inefficient metabolism and thermogenesis. Consider:
- UCP3 is mainly expressed in muscle tissue.6
- UCP3 is down-regulated during weight loss in humans.3
- UCP3 is increased in muscle tissue by leptin.7
- UCP3 causes mitochondrial uncoupling when expressed in yeast.7
- UCP3 expression is correlated with inefficient metabolism in real, walking-around humans.8
- UCP3 polymorphisms are associated with obesity in real, walking-around humans.9,10
The UCP3 Thermogenesis Controversy
Despite all of this evidence it is still controversial to suggest that UCP3 has any kind of thermogenic role in mammals! Soon after the discovery of UCP3 it was found that mice who lack UCP1 cannot do the rapid thermogenesis in response to cold even though they have a functional UCP3 gene. This led to a flurry of articles such as this one:
UCP1 : the ONLY protein able to mediate adaptive non-shivering thermogenesis and metabolic ineffciency
The uniqueness of UCP1 (as compared to UCP2/UCP3) is evident from expression analysis and ablation studies. UCP1 expression is positively correlated with metabolic inefficiency, being increased by cold acclimation (in adults or perinatally)and overfeeding, and reduced in fasting and genetic obesity. Such a simple relationship is not observable for UCP2/UCP3. Studies with UCP1-ablated animals substantiate the unique role of UCP1 : the phenomenon of adaptive adrenergic non-shivering thermogenesis in the intact animal is fully dependent on the presence of UCP1, and so is any kind of cold acclimation-recruited non-shivering thermogenesis ; thus UCP2/UCP3 (or any other proteins or metabolic processes) cannot substitute for UCP1 physiologically, irrespective of their demonstrated ability to show uncoupling in reconstituted systems or when ectopically expressed
Nedergaard, et al.
It rapidly became unfashionable to suggest that UCP3 was involved in thermogenesis. The study where Rosenbaum et al. showed that UCP3 was reduced by 28% when subjects lost weight was done in 1999. The paper suggesting that UCP1 was the ONLY UCP involved in thermogenesis came out in 2001. The study where they showed that leptin restored the metabolic rates and inefficient metabolism of dieters – the paper I led with – was done in 2005, again by Rosenbaum et al. In 1999 they were interested in UCP3, in 2005 they were not. They SHOULD have been curious about UCP3 levels before weight loss, after weight loss and then again after leptin injection, they had the know how. They lost their curiosity. Peer pressure? Group think? Probably. A small tragedy.
UCP3 Driven Mammalian Thermogenesis Makes a Comeback
Then in 2016 this came out,11 concluding that “both uncoupling protein 1 (UCP1) and UCP3 are important for mammalian thermoregulation.” The authors compared the thermogenic responses of wild type mice, mice lacking UCP1, mice lacking UCP3 and mice lacking both in multiple contexts that would lead to thermogensis – cold, bacterial challenge and methamphetamine. And they made pretty pictures! I’ll start with methamphetamine because that image is the most illustrative.
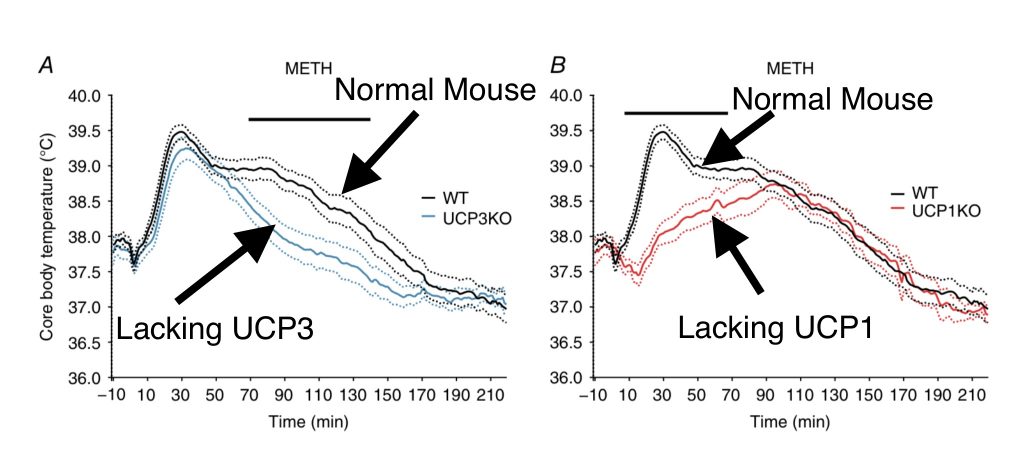
In mice injected with methamphetamine, there is a quick spike in body temperature that is induced by UCP1. In mice lacking UCP3 (blue line) body temperature spikes and then quickly falls. In mice lacking UCP1 (red line), the quick spike in body temperature doesn’t happen, but body temperature continues to rise over time and stays elevated longer. If you add up the blue line – UCP1 induced thermogenesis – and the red line – UCP3 induced thermogenesis – you get the same curve as in wild type mice. UCP1 induced thermogenesis in brown adipose tisuue comes on very quickly and very intensely. UCP3 induced thermogenesis in skeletal muscle is a slower starting, less intense but longer lasting (in this case) thermogenesis.
But clearly UCP3 is playing an important role in meth induced thermogenesis!
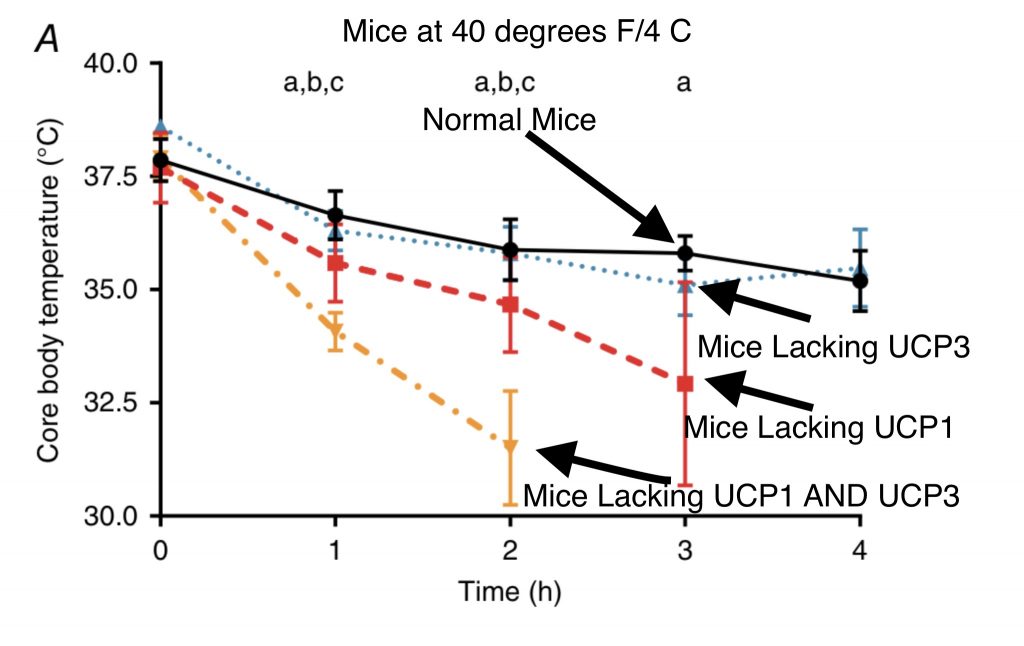
The authors also considered the earlier claim that UCP3 is NOT involved in cold-induced thermogenesis by comparing normal mice kept at 40 degrees F (4 degrees C) Vs mice without UCP1, mice without UCP3 and mice without BOTH. The results show that the earlier authors had it wrong. In mice, the UCP1 induced response to cold is SUFFICIENT to maintain relatively high body temperature. Just because UCP1 is sufficient doesn’t mean that UCP3 isn’t contributing. In fact, mice lacking both UCP1 and UCP3 have a much faster drop in body temperature than mice lacking only UCP1. UCP3 is clearly playing a role that is masked by UCP1 in normal mice.
Conclusion
Consider a pot of tea and a teacup of tea. The teacup has a much higher surface to volume ratio that the pot and it cools off much quicker. The mouse is the teacup. Mice are in danger of RAPID body temperature drop in response to cold. Therefore, they have a relatively high proportion of Brown Adipose Tissue and UCP1. They need a robust, quick response. Humans are the pot of tea. We can handle 40 degrees for two hours, it’s fine. We may not like it, but we won’t die. We run our thermogenesis through UCP3 in skeletal muscle. It takes a little longer and that’s OK.
I believe that increased UCP3 expression in skeletal muscle is the target of leptin induced thermogenesis in humans. This is the simplest answer to the questions of why metabolic rate drops disproportionately in weight loss, why leptin restores it, why UCP3 is associated with inefficient metabolism and why leptin polymorphisms are associated with obesity.
- 1.Rosenbaum M. Low-dose leptin reverses skeletal muscle, autonomic, and neuroendocrine adaptations to maintenance of reduced weight. Journal of Clinical Investigation. Published online December 1, 2005:3579-3586. doi:10.1172/jci25977
- 2.Rosenbaum M, Murphy EM, Heymsfield SB, Matthews DE, Leibel RL. Low Dose Leptin Administration Reverses Effects of Sustained Weight-Reduction on Energy Expenditure and Circulating Concentrations of Thyroid Hormones. The Journal of Clinical Endocrinology & Metabolism. Published online May 1, 2002:2391-2394. doi:10.1210/jcem.87.5.8628
- 3.Vidal-Puig A, Rosenbaum M, Considine RC, Leibel RL, Dohm GL, Lowell BB. Effects of Obesity and Stable Weight Reduction on UCP2 and UCP3 Gene Expression in Humans. Obesity Research. Published online March 1999:133-140. doi:10.1002/j.1550-8528.1999.tb00694.x
- 4.Klaus S, Casteilla L, Bouillaud F, Ricquier D. The uncoupling protein ucp: A membraneous mitochondrial ion carrier exclusively expressed in brown adipose tissue. International Journal of Biochemistry. Published online January 1991:791-801. doi:10.1016/0020-711x(91)90062-r
- 5.Lowell BB, S-Susulic V, Hamann A, et al. Development of obesity in transgenic mice after genetic ablation of brown adipose tissue. Nature. Published online December 1993:740-742. doi:10.1038/366740a0
- 6.Schrauwen P, Hesselink M. UCP2 and UCP3 in muscle controlling body metabolism. Journal of Experimental Biology. Published online August 1, 2002:2275-2285. doi:10.1242/jeb.205.15.2275
- 7.Gong D-W, He Y, Karas M, Reitman M. Uncoupling Protein-3 Is a Mediator of Thermogenesis Regulated by Thyroid Hormone, β3-Adrenergic Agonists, and Leptin. Journal of Biological Chemistry. Published online September 1997:24129-24132. doi:10.1074/jbc.272.39.24129
- 8.Schrauwen P, Troost F, Xia J, Ravussin E, Saris W. Skeletal muscle UCP2 and UCP3 expression in trained and untrained male subjects. Int J Obes. Published online September 1999:966-972. doi:10.1038/sj.ijo.0801026
- 9.Salopuro T, Pulkkinen L, Lindström J, et al. Variation in the UCP2 and UCP3genes associates with abdominal obesity and serum lipids: The Finnish Diabetes Prevention Study. BMC Med Genet. Published online September 21, 2009. doi:10.1186/1471-2350-10-94
- 10.Liu Y-J, Liu P-Y, Long J, et al. Linkage and association analyses of the UCP3 gene with obesity phenotypes in Caucasian families. Physiological Genomics. Published online July 14, 2005:197-203. doi:10.1152/physiolgenomics.00031.2005
- 11.Riley CL, Dao C, Kenaston MA, et al. The complementary and divergent roles of uncoupling proteins 1 and 3 in thermoregulation. J Physiol. Published online November 13, 2016:7455-7464. doi:10.1113/jp272971

so, hear me out — could supplementation with niacin/nicotinic acid boost the effectiveness of the leptin we produce by reducing the level of triglycerides in the bloodstream? could the flush be activating more than just the tingles? it definitely feels like my body temp skyrockets when I take it. curious to get your thoughts, although I’m not sure flush niacin jives very well with your ROS theory — or maybe it does?
I honestly don’t know much about niacin flushing.
It probably does. Niacin upregulates UCP1 (in mice)
https://www.biorxiv.org/content/10.1101/382416v1.full
See figure 6c.
Brilliant as always Brad. Have you looked in to the effects of Betulinic acid (found in Chaga) and/or Magniferin (mango leaf extract) on SCD1/SREBP1? Both seem to be potent supressors.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3943915/
https://pubmed.ncbi.nlm.nih.gov/26647913/
I find the link between SCD1 and cancer interesting too.
Interesting… I haven’t had time to look them up but thanks for sharing!
Hi, I just started following your work and I’m finding it really interesting. This article was good. Have you heard of Ray Cronise and his winter hypothesis? Mr. Cronise was working on a lot of the same ideas you have been writing about. He was working on a book about the history of diet around the early 1900’s. I’m starting on your diet and I hope to see some good results. Thanks for your work.
No, I hadn’t heard of him. You guys are full of interesting suggestions!
Great article. I don’t understand all of it but enough to say this may be the first nail in the coffin of CICO orthodoxy.
People living in equatorial regions tend to be lean and ancestrally wouldn’t have needed to store fat for the winter. I suspect UCP3 expression is dependent on the prevalent climate and seasonal foods. It’s very likely that there are other ways to burn fat or metabolize it away other than thermogenesis. Interesting hypothesis though.
So what’s the usual turnaround Time on orders? I ordered last week and haven’t seen a confirmation. I’m not irritated or anything but am very eager to get started. Just knowing about the time frame would help. Cheers
Usually they go out very quickly. There has been a mixup at the warehouse this week. I am working on the problem and I expect everything to ship today. Sorry about the delay.
Brad
Leptin and inefficiency made me think of Huntington’s disease, and the terrible weight loss that can result. A quick search brought me to this: https://pubmed.ncbi.nlm.nih.gov/19548952/
Interesting. There’s a lot to unpack there.
So… if it’s mostly in our muscles that deal with cold… is that why we shiver? The muscles tremble, is that doing something related to it?
Shivering is actually a separate thing. In the literature the kind of thermogenesis I’m taking about is called “non-shivering thermogenesis”. SO yes, shivering DOES lead to thermogenesis, but it’s not the only way.