In the last article we saw that a deficiency in AMPK and SIRT1 activity leads to the inactivation of the mitochondrial SIRT3 deacetylase, acetylated (turned off) mitochondrial enzymes and low metabolic rate. I suggested at the end that to escape this pattern, you have to activate AMPK and SIRT1 at the same time. In this article I’ll give you a little more background on PGC-1a and SIRT1 and look at a dietary trial to see the effects of hi-fat or lo-fat overfeeding and underfeeding on SIRT1-AMPK-PGC-1a.
PCG-1a is the master regulator of mitochondrial biogenesis
PGC-1a plays an outsized role in metabolic rate. It is said to be the master regulator of mitochondrial biosynthesis and energy expenditure.1 It is crucial in the differentiation of brown adipose tissue, a major thermogenic organ.2 Brown adipose tissue is brown due to it’s high levels of mitochondria! PCG-1a is also involved in the browning and therefore thermogenic and oxidative role of white fat cells.3 The white fat tissue of obese humans has a lower thermogenic rate than white adipose tissue from lean humans.
Additionally, PGC-1a plays a major role in determining muscle fiber composition.4 The major muscle fibers are type I, IIa and IIb. Obese humans have proportionally more Type IIb fibers and less type I and type IIa fibers.5 Type I fibers are “slow twitch”, have a high mitochondrial content and are dark red. Type IIa fibers are fast twitch but with high oxidative capacity and high levels of mitochondria. Type IIb fiber are “white muscle” – think of the light colored pork loin in a pork chop. White muscle fibers are called “glycolytic” – they derive their energy from glycolysis and produce lactic acid. They have low oxidative potential and tire more quickly. Overexpressing PGC-1a in the muscle of pigs or mice leads the muscle fiber to be much more oxidative.4
SIRT1 and AMPK co-activate PGC-1a and SIRT3
To be activated, PGC-1a has to be both phosphorylated by AMPK and deacetylated by SIRT1. When PGC-1a is activated, it increases expression of SIRT3 and SIRT3 is activated by being deacetylated by SIRT1. Activated SIRT3 deacetylates your mitochondrial enzymes and takes the breaks off of your metabolic rate.
Hibernating animals go through a cycle of states every two weeks or so. Entrance into torpor, early torpor, then late torpor followed by two stages of arousal. In late torpor – in anticipation of arousal and the coming burst of thermogenesis it will take to raise their body temperature from almost freezing to warm blooded – hibernating squirrels massively increase SIRT3 expression in skeletal muscle.6

Lean humans can turn on AMPK and SIRT1 by feasting on fatty food
This paper7 did a trial on overfeeding lean humans (average BMI 21.8) on a lo-fat (20% calories from fat, 60% carb) or hi-fat (50% fat, 30% carb) diet. Baseline caloric consumption was determined for each person and they were fed that number of calories for five days as 30% fat calories and 50% carbohydrate. Then they were fed 140% of that amount for 5 days on either the high-fat or lo-fat diet. Muscle tissue biopsies were taken to determine relative pAMPK and deacetylated PGC-1a levels, the activated forms of both. This was a “crossover” trial where you measure baseline values of each person and then again after the different treatments. There was a 30 day “washout” between the different diets where the participants just ate their normal diet. It’s a good design because each person serves as their own internal control.
Here are the results.
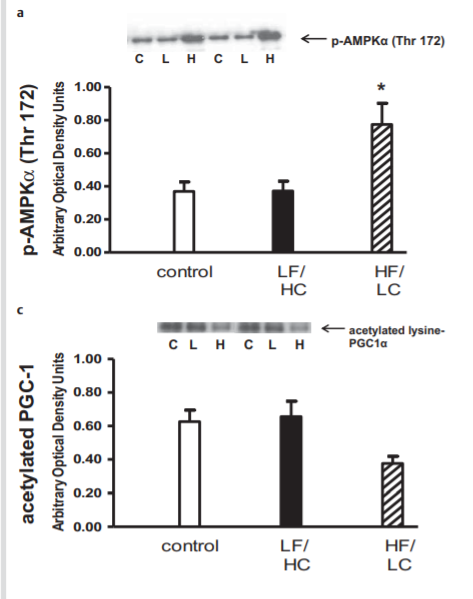
I think of this experiment as “The French Christmas Experiment”. Very lean volunteers responded to short term, hi-fat overfeeding by upping their rates of fat oxidation by activating both AMPK and SIRT1. Think of Buche de Noel and the fatty traditional Christmas boudin blanc sausages in France. People who are metabolically healthy make it through holiday feasting without becoming obese.
I am amused by this result since the language around AMPK and SIRT1 is typically the puritanical language of the calories-in, calories-out school of obesity thought. “SIRT1 in turn is best known historically for producing genetic changes that mediate the increase in longevity caused by calorie restriction.”7 Apparently feasting on fat also works.
Obese humans can turn off AMPK and SIRT1 with a low-cal, low-fat diet
The research group repeated the experiment with obese adults and calorie restriction. They put obese humans on a 30% calorie reduced diet on the same macro ratios. I would love to have seen lean humans on calorie reduction and obese humans on overfeeding as well but no such luck. Here are the results of that trial.
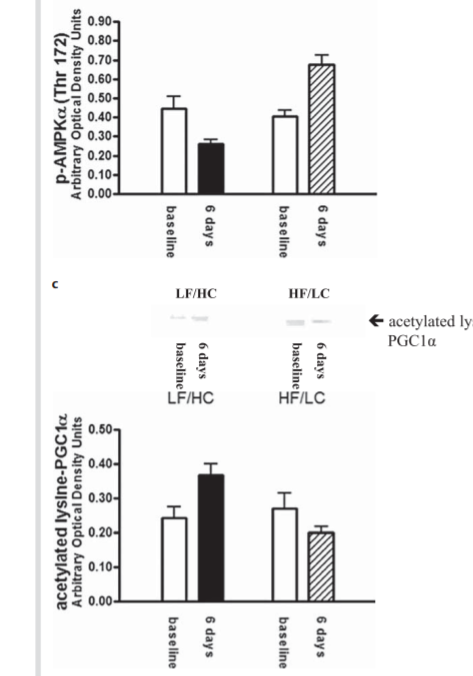
The reduced calorie hi-fat diet increased AMPK and SIRT1 activation. The lo-fat diet did the opposite. If you’re going to restrict calories, do it with a fatty diet!
Let’s look at how the macros actually changed in these two experiments. I’m using 2400 calories per day as a starting point and I’m particularly interested in the overfed hi fat diet (the most effective one) and the low calorie, lo-fat diet (the worst one).
| Isocaloric | Overfed Hi Fat | Calorie Restricted Hi Carb | |
| Calories | 2400 | 3360 | 1680 |
| Protein | 120g | 168g | 84g |
| Carbohydrate | 300g | 252g | 252g |
| Fat | 80g | 224g | 38g |
| Effect On AMPK/SIRT1 | Control | UP | DOWN |
The authors amazingly conclude, “Our data indicate that a relative deficiency in carbohydrate intake or, albeit less likely, a relative excess of fat intake even in the absence of caloric deprivation is sufficient to activate the AMPK-SIRT1-PGC1α energy-sensing cellular network in human skeletal muscle.” This is despite the fact that the overfed hi-fat group and the calorie restricted hi-carb group had the EXACT same amount of carbohydrate reduction and OPPOSITE effects on AMPK-SIRT1-PGC-1a.
The authors didn’t have the heart to write the actual conclusion: the standard advice given to the obese – restrict calories and avoid fat – had the worst effect on the AMPK-SIRT1-PCG1a axis. Feasting on fat did the opposite.
- 1.Fernandez-Marcos PJ, Auwerx J. Regulation of PGC-1α, a nodal regulator of mitochondrial biogenesis. The American Journal of Clinical Nutrition. Published online February 2, 2011:884S-890S. doi:10.3945/ajcn.110.001917
- 2.Kang S, Bajnok L, Longo KA, et al. Effects of Wnt Signaling on Brown Adipocyte Differentiation and Metabolism Mediated by PGC-1α. Mol Cell Biol. Published online February 15, 2005:1272-1282. doi:10.1128/mcb.25.4.1272-1282.2005
- 3.Fisher f. M, Kleiner S, Douris N, et al. FGF21 regulates PGC-1 and browning of white adipose tissues in adaptive thermogenesis. Genes & Development. Published online February 1, 2012:271-281. doi:10.1101/gad.177857.111
- 4.Zhang L, Zhou Y, Wu W, et al. Skeletal Muscle-Specific Overexpression of PGC-1α Induces Fiber-Type Conversion through Enhanced Mitochondrial Respiration and Fatty Acid Oxidation in Mice and Pigs. Int J Biol Sci. Published online 2017:1152-1162. doi:10.7150/ijbs.20132
- 5.MArin P, Andersson B, Krotkiewski M, Bjorntorp P. Muscle Fiber Composition and Capillary Density in Women and Men With NIDDM. Diabetes Care. Published online May 1, 1994:382-386. doi:10.2337/diacare.17.5.382
- 6.Rouble AN, Storey KB. Characterization of the SIRT family of NAD+-dependent protein deacetylases in the context of a mammalian model of hibernation, the thirteen-lined ground squirrel. Cryobiology. Published online October 2015:334-343. doi:10.1016/j.cryobiol.2015.08.009
- 7.Draznin B, Wang C, Adochio R, Leitner J, Cornier M-A. Effect of Dietary Macronutrient Composition on AMPK and SIRT1 Expression and Activity in Human Skeletal Muscle. Horm Metab Res. Published online June 6, 2012:650-655. doi:10.1055/s-0032-1312656

wow that’s incredible. It also aligns with my real world experience. Been dieting since I was 18 (I’m 31 now) eventually got to a point where I was GAINING weight eating 1100 calories/day. People told me I must have been estimating the calories wrong or forgetting to log mindless eating/sneaking food etc. I KNEW what I ate and I thought it was a cruel joke. Then I discovered keto, and lost 50 lbs eating 2400 or so calories a day of mostly saturated fat. The only problem is once I stopped I gained 15 lbs back and haven’t been able to lose those 15 lbs again no matter what diet I do. I’ve tried carnivore, TCD, psmf, etc. Now I’m supplementing with berberine and other ampk activators. I’m hoping your research finally helps me and everyone else who’s been stuck like me figure it all out.
Hey Ryan. I assume you transitioned from strict keto because you weren’t doing well on it long term? Seems like folks are coming out of the woodwork lately with horror stories of how keto, carnivore etc. ended up causing metabolic damage or just generally poor health outcomes. Suddenly seeing a lot more talk of “pro-metabolic” eating in the health sphere at large.
I’ve been low carb for years but am trying to transition back to more balanced macros. I got a little ahead of myself at first and definitely gained more than I was comfortable with. Kind of starting over at the moment and trying to add back carbs as slowly as I can.
What are your macros like now, and outside of the unwanted 15 lbs, how do you feel mentally and physically with your current diet? A lot of the pro-metabolic folks are quick to point out that even with a measured approach, weight gain when transitioning back to more balanced macros is almost inevitable, and eventually your body will reach a kind of homeostasis w/ the glucose and your weight should stabilize.
yeah, lo cal low fat, all that stuff ruined me, and i got so fat after i went off it… so i went keto about this time last year, and lost 30 lbs sloooowly, then gd tim ferris talked me into ‘cheat days’… i gained 14 lbs after 2 cheat days, when back on keto and it wouldn’t work for me anymore… so i said a prayer and found Brad, but i think i’m in deeeep torpor
Interesting …. So that’s the keto experience eh? Triggering AMPK-SIRT1-PGC-1a by eating a bunch of fat.
Also find the muscle fiber discussion interesting- Dr. Ben Bocchiccio’s 15 minutes to fitness plan ( http://drbenbo.com/ ) I learned from LowCarb MD podcast involves, basically, doing high-intensity slow exercises to invoke enough load, for long enough, to trigger type IIb muscle fibers and exhaust them. It’s specifically designed to get you to safely access those muscle fibers and burn off their glycogen. I hate doing it, and in fact keep skipping my workouts, need to get back into it. But it’s really nice when I can get my fatass into the mood to do it.
This makes perfect sense. Low carb environment would create a situation where more mitochondria are needed to go produce energy from existing fat stores. This is also why exercise can upregulate PGC1a, it’s mainly a response to the nutrient depletion.
For those that like to supplement PQQ (usually combined with CoQ10) is an activator of PGC1a.
what was the SFA/MUFA/PUFA composition of the “Overfed Hi Fat” diet?
From the study
“and the HF/LC diets were enriched in dairy, nuts, and oils”
“Dietary lipid for all diets contained a 1:1:1 ratio of
monounsaturated, polyunsaturated, and saturated fats.”
So 60% fat from the diet, 1/3 of that being polyunsaturated fat, Is 20% of calories from PUFA.
That sounds right. In other studies, there is a quick increase in SIRT1/3 levels due to increasing unsaturated fat levels which reverses over time. I think the reversal is prob dependent on saturation level.
Unfortunately it wasn’t reported.
Do we have any data on how the levels of SIRT3 correspond to overall metabolic rate ?
(Presumably it’s high enough to partly explain the difference between someone being lean and obese)
Can we compare between the two graph’s, because the calorie restricted group has “better” numbers, but the Y-axis is labelled suspiciously?
I don’t think you can compare between the graphs in an experiment like this. You have a gel with radioactive bands. The darkness of the bands depends on how long you expose the film. So in the last graph they may just have not exposed the film as long.
Hi Everyone,
Lee here from the UK. I have really been enjoying all of this journey that Brad is taking us on over the last 6 months, trialling a few of the tips given along the way. I firmly believe Brad is on to something here so I really stepped things up during the last 2 months and I am now seeing fairly consistent results so I thought I would finally give you all my thoughts / tactics on it all.
So my own tactics are the following:
Me: So I weighed 19.5 stone 2 months back with a fair bit of muscle but the bulk of my fat around my belly area. I would estimate a body fat of 25% but that is an estimate. I am slightly over 6ft, strength train as many days as I can each morning in a fasted state and work in an office 8-5 Mon to Fri.
Diet: So I have settled on kind of a cross breed between Brads Croissant Diet, Ted Naiman’s Protein Energy Diet all while trying my best to follow Bill Schindlers advice about making foods more digestible (fermentation / sourdough etc). Meat & Fish are the focal point of my diet with me taking in about 300g of protein a day via 1.2kg / 1.3kg of meat/ fish. My carb intake is usually around 100g a day which I usually have during my last meal of the day. My fat intake is usually around 100g a day which I focus on Saturated fat sources (Beef Suet / Cheese / Cream / Butter / naturally from the meats/ fish). In general I choose lean meats and fish (Hanger steak over rib eye / Chicken Breast over Legs / Trimmed Pork Loin over Belly for example). My carb sources are usually a mix of starch and a bit of sugar (Example – A small portion of Mashed Potatoes with my dinner followed by a small dessert of chocolate brownie or a small sourdough scone with Jam)
Results so far: So a few months back I tried a much higher fat intake of more like 250g – 300g and could feel the burn which resulted in me not losing any weight but also not gaining weight despite the extra 1800 calories a day I was taking in. So for certain the higher saturated fat increase works but it wasn’t enough originally to get me moving in the right direction despite a few months of trying it. I swapped 2 months back to my new diet explained above with limited carbs and fat (energy) while prioritising protein, combined with my supplements listed below and performing high intensity weight training each morning in a fasted state (5-6 days a week). This combo has finally started to shift my weight with me now weighing in at 18.10 but I am almost certain I am putting muscle on while this is happening. I am very rarely hungry so I feel this is easily sustainable and my long term game plan is to slowly increase my saturated fat intake bit by bit but only as I drop total body fat. If my weight loss stalls then I will simply drop the fat back down until it starts again. My belief is that I will slowly drop body fat and this will all become easier as I go with my current tactics but I will look to update you all again in 3 months time to tell / maybe even show my results.
Supplements: Berberine (2/3g a day) / Sterculia Oil (3 Squeezes a day) / Astaxanthin (12g a day) / Vitamin D 3 + K2 (1 tab a day) / Iodine (22mg a day) / Resveratrol (250mg Twice a day)
Questions: Brad, could you please give me your opinion on the amounts that I am currently taking my supplements in and your thoughts on the timing of these each day?? Any obvious tweaks you can suggest to my current game plan??
Apologies for what seems like a giant post to me but I hope this helps or is at least of interest to you all. I look forward to sharing my journey as I go, your comments to this post and thank you again Brad for your efforts in breaking all of this down for us.
Thanks for sharing! Very interesting! I think your supplements seem reasonable, perhaps I would do 2 squeezes on the Sterculia Oil, there’s definitely a sweet spot before you start to see inflammatory numbers rising.
SFA:MUFA:PUFA 1:1:1 for all diets
Does your theory explain why zoo animals are getting fatter too despite isocaloric chow since the 70s? Or are micro-plastics still a decent hypothesis for why obesity is increasing?
(https://psmag.com/social-justice/just-people-getting-fatter-65342)
There are a lot of reasons this could happen. One is a generational handdown of increased SCD1 activity via cord blood/mother’s milk (in the case of mammals). But yes, enviromental toxins certainly also play a role and are probably signalling via the nuclear receptor superfamily, including AhR, SXR, etc. I haven’t talked about it on the blog yet, cause I didn’t want to go TOO far down the nuclear receptor rabbit hole, but the SXR behaves similarly to the AhR and is triggered by plastics.
http://citeseerx.ist.psu.edu/viewdoc/download?doi=10.1.1.623.7582&rep=rep1&type=pdf
Wouldn’t it be best to Decrease AMPK/SIRT while trying to lose weight and in a caloric deficit and to increase MTOR in order to spare lean mass during weight loss?
In the sense that AMPK/SIRT help you burn fat I can’t see the logic of wanting to reduce them.