I’ve been working on this mega post that was getting too long and complicated. I decided to break it into three or four parts and start at what was going to be the end. I’m feeling really excited about this series of posts because I feel like we’re really getting to the root causes of obesity and metabolic syndrome.
I know the molecular biology has gotten pretty deep on Fire In A Bottle as of recent. Hold on for this series, it’s important! Then I have some higher level articles that won’t be quite as heady and that I think will be thought provoking.
In this mini-series we’re going to add three new characters – important ones: the sirtuins SIRT1 and SIRT3 and the transcriptional co-activator PGC-1a. I’ll introduce SIRT3 in this article and SIRT1 and PGC-1a in the next one.
In this article we’ll see that SIRT3 levels are low in the adipose tissue of obese children and that the enzymes involved in oxidative metabolism have been turned off by a post-translation type of regulation called acetylation.
Post Translational Regulation
Many enzymes are regulated by a process called “post-transational regulation”. This means that enzymes can be produced (translated) and are in the cell, but are not necessarily active until certain conditions are met. One of the common forms of post-translation regulation are enzymes that attach commonly available molecules to the protein itself. Kinases, such as AMPK (AMP Kinase) – the hero of my latest series of posts – are a good example of this. Kinases attach phosphate groups to enzymes. Sometimes this turns the enzyme ON and sometimes it turns it OFF. It depends. Conversely, phosphatases REMOVE phosphate groups. So the activity level of an enzyme such as ACC, which plays a crucial role in switching between fat burning and glucose burning and which is turned OFF by phosphorylation, is determined by the level of AMPK which phosphorylates it and a phosphatase which dephosphorylates it. Got it?
Phosphate groups are obtained through the diet, well absorbed and water soluble. They literally just float in solution until a kinase decides to stick them onto something and turn it on. Or off. It depends.
Phosphorylation is the most well known and well studied form of post translational regulation, but as the years have gone by we’ve discovered more and more types of post-translational regulation and realized their importance. This is true of an hilarious number of small molecules. Let’s see, we have methylation, propionylation and butyrylation(discovered in 2007)1, biotinylation, succinylation (2010)2, malonylation(2011)3, alkylation, gamma-carboxylation (Vitamin K dependent). Basically, anything that was lying around, enzyme systems evolved that sticks them on and takes them off of enzymes as a switch.
Acetylation is a type of post-translational modification that is controlled by acetyltransferases and deacetylases. Humans have a family of deacetylases called the sirtuins. There are 7 mammalian sirtuins4, abbreviated as SIRT1-SIRT7. Sirtuins initially became of interest because they were found to be crucial for extending the lifespan of calorie restricted yeast. They are broadly conserved and their level of activity depends on the ratio of NAD+/NADH. In this way they are “energy sensors”, similar to AMPK.
The level of acetylation of most human enzymes is controlled by the level of the sirtuins. SIRT3 localizes to the mitochondria and controls the level of mitochondrial enzyme acetylation.
The mitochondrial proteins of obese humans are highly acetylated
A 2018 Nature paper showed the shocking degree of acetylation of mitochondrial enzymes in adipose tissue of obese children5, including the enzymes citrate synthase and complex 1. Oxidative metabolism is controlled by the citric acid cycle, which converts acetyl-CoA derived from either glucose, fat, ethanol, protein, or ketones into FADH2 and NADH. The NADH and FADH2 are then passed through the mitochondrial electron transport chain. The first step of the citric acid cycle – adding a new acetyl group (ironically) onto oxaloacetate to form citrate – is catalyzed by citrate synthase.
The NADH produced during the citric acid cycle then gets passed into complex I of the electron transport chain. When the electrons flow through the chain, they are eventually combined with O2 to make water. Oxygen is the terminal electron acceptor and that’s why we call it oxidative metabolism.
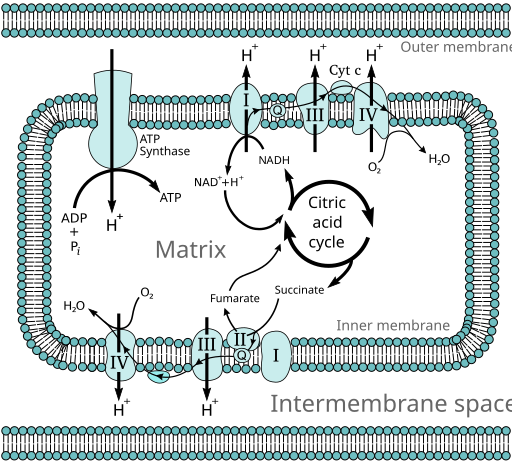
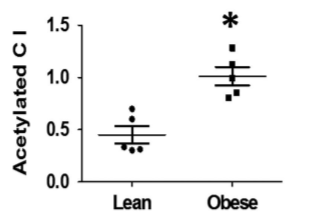
Adipose (fat) tissue of obese children has low levels of SIRT3.

In the mitochondria of obese children, citrate synthase and complex I are highly acetylated and turned OFF.
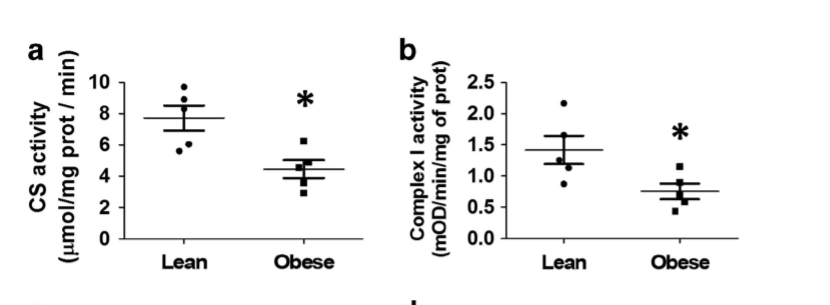
You can see the problem. How can an obese person efficiently do oxidative metabolism if their enzymes are turned off? In addition to Complex I and Citrate synthase being turned off, Complex V – the one that makes the ATP – is also hyperacetylated and ATP levels are low.
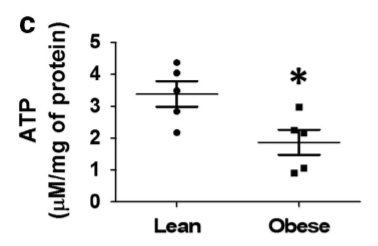
It has long been known that both the fat tissue6 and muscle tissue7 of obese humans have low oxygen consumption rates. I suspect that acetylated mitochondrial enzymes play a large role in this.
Regulating SIRT3
The tissues of obese humans have low oxygen consumption rates, highly acetylated enzymes involved in oxidative metabolism and therefore, presumably, low levels of SIRT3 activity. How is SIRT3 activated? That’ll be the subject of the next post, but I’ll give you the brief version.
SIRT3 is activated by being deacetylated by the deacetylase SIRT1.8 SIRT1 is turned on by being phosphorylated by AMPK9. The amount of SIRT3 can be increased by the transcriptional co-activator PGC-1a10. PGC-1a increases expression of itself.11 PGC-1a is activated only when it is both deaccetylated by SIRT1 and phosphorylated by AMPK.12
Did you get all of that? The take home is that to decrease mitochondrial enzyme deacetylation you have to stimulate AMPK and SIRT1 at the same time. I have a lot more to say about how to do this, but if you want to try something in the short term, resveratrol activates SIRT1 and berberine activates AMPK. You could try taking them together.
- 1.Chen Y, Sprung R, Tang Y, et al. Lysine Propionylation and Butyrylation Are Novel Post-translational Modifications in Histones. Molecular & Cellular Proteomics. Published online May 2007:812-819. doi:10.1074/mcp.m700021-mcp200
- 2.Zhang Z, Tan M, Xie Z, Dai L, Chen Y, Zhao Y. Identification of lysine succinylation as a new post-translational modification. Nat Chem Biol. Published online December 12, 2010:58-63. doi:10.1038/nchembio.495
- 3.Peng C, Lu Z, Xie Z, et al. The First Identification of Lysine Malonylation Substrates and Its Regulatory Enzyme. Molecular & Cellular Proteomics. Published online December 2011:M111.012658. doi:10.1074/mcp.m111.012658
- 4.Haigis MC, Guarente LP. Mammalian sirtuins–emerging roles in physiology, aging, and calorie restriction. Genes & Development. Published online November 1, 2006:2913-2921. doi:10.1101/gad.1467506
- 5.Zamora-Mendoza R, Rosas-Vargas H, Ramos-Cervantes MT, et al. Dysregulation of mitochondrial function and biogenesis modulators in adipose tissue of obese children. Int J Obes. Published online November 21, 2017:618-624. doi:10.1038/ijo.2017.274
- 6.Böttcher H, Fürst P. Decreased white fat cell thermogenesis in obese individuals. Int J Obes. Published online June 1997:439-444. doi:10.1038/sj.ijo.0800425
- 7.Hulver MW, Berggren JR, Carper MJ, et al. Elevated stearoyl-CoA desaturase-1 expression in skeletal muscle contributes to abnormal fatty acid partitioning in obese humans. Cell Metabolism. Published online October 2005:251-261. doi:10.1016/j.cmet.2005.09.002
- 8.Kwon S, Seok S, Yau P, Li X, Kemper B, Kemper JK. Obesity and aging diminish sirtuin 1 (SIRT1)-mediated deacetylation of SIRT3, leading to hyperacetylation and decreased activity and stability of SIRT3. Journal of Biological Chemistry. Published online October 2017:17312-17323. doi:10.1074/jbc.m117.778720
- 9.Lee C-W, Wong LL-Y, Tse EY-T, et al. AMPK Promotes p53 Acetylation via Phosphorylation and Inactivation of SIRT1 in Liver Cancer Cells. Cancer Res. Published online June 22, 2012:4394-4404. doi:10.1158/0008-5472.can-12-0429
- 10.Ansari A, Rahman MdS, Saha SK, Saikot FK, Deep A, Kim K-H. Function of the SIRT3 mitochondrial deacetylase in cellular physiology, cancer, and neurodegenerative disease. Aging Cell. Published online September 29, 2016:4-16. doi:10.1111/acel.12538
- 11.Joseph A-M, Pilegaard H, Litvintsev A, Leick L, Hood DA. Control of gene expression and mitochondrial biogenesis in the muscular adaptation to endurance exercise. Wagenmakers AJM, ed. Essays in Biochemistry. Published online November 27, 2006:13-29. doi:10.1042/bse0420013
- 12.Cantó C, Jiang LQ, Deshmukh AS, et al. Interdependence of AMPK and SIRT1 for Metabolic Adaptation to Fasting and Exercise in Skeletal Muscle. Cell Metabolism. Published online March 2010:213-219. doi:10.1016/j.cmet.2010.02.006

Neat, so now we’re getting into what AMPK does.
That mitochondrial enzymes acetylate in obesity/torpor makes sense – Gotta switch off as many unnecessary “cylinders” from the engine as you can get away with to survive the famine (or winter).
So if I got things right, now I should take my berberine with red wine?
I’m starting to like you a lot Brad!
The amount that’s actually in wine is actually very small, and is not likely to be at a therapeutic level.
https://usualwines.com/blogs/knowledge-base/resveratrol-wine
>Furthermore, dark red and purple grapes have a higher concentration of resveratrol than other grape varieties. As such, you’ll find that Malbec, Syrah, Pinot Noir, and Cabernet Sauvignon tend to have the highest resveratrol levels.
>That said, resveratrol wine amounts are not as high as you might think. Studies show that resveratrol concentrations in wine can range from negligible to around 1.979 milligrams per liter (for Pinot Noir from France). Research also shows that you’d likely need to consume at least 1 gram of resveratrol (1,000 milligrams) daily to experience any of the significant therapeutic benefits it offers.
>To put these figures into perspective, that means you’d have to consume more than 505 liters — a whopping 133 gallons! — of Pinot per day to get the health benefits. Yes, you read that right. (We’ll wait while you pick up your jaw from the floor.)
>Of course, nobody would ever recommend such an insane consumption of red wine, but it just goes to show that the health benefits of resveratrol have been a bit overblown when measured by realistic standards.
Even Brad would have a hard time getting enough from wine “naturally”. 🙂
Tx for doing the math. I’ll stick with my 1L/week red wine.
Hoping homeopathic doses will do!
I’m sticking to sterculia oil + berberine combo until my next OmegaQuant test.
I was just trying to show I am (somewhat) following despite the fact I have lost hope of understanding the biology science of this work!
Is this a challenge?
You could do worse.
In an earlier post you mentioned that berberine was giving some people issues and it seemed like you were now in favor of using Astaxanthin instead. Do they both do similar things or is berberine doing something different from Astaxanthin in this case?
IIRC astaxanthin was doing something different with PPARg inhibition and PPARa agonism. There was something else going on that Brad mentioned about astaxanthin parking itself inside the mitochondrial membrane or something. Now with sirtuins playing a part in the story I wonder what astaxanthin is really doing.
PS- Berberine is one of a staggering number of AMPK agonists.. Jiaogulan tea, Hibiscus (my current experiment), Gentian root/campari/bitters, anthocyanins from blueberries, teas (incl EGCG?) et al, lots of flavonols and polyphenols seem to play this part. I am starting to think of AMPK agonists as the springtime anti-torpor tonic, and just look to things that grow in springtime for rough guidance.
Side note- Dandelions are a typical sight here in spring and often heralds the truest signal that “spring is here”. Lo and behold: https://pubmed.ncbi.nlm.nih.gov/29234282/
Dandelions have that very bitter flavor. My guess is they are triggering bile acid receptors to increase pAMPK.
So lots and lots of French Burgundy and dandelion wine? I picked the wrong week to quit drinking.
BTW I started taking 12mg of astaxanthin twice a day a few weeks ago. Can anyone thing of why this would cause my heart arrhythmia to go away? The effect was literally overnight.
No idea but that is cool! Makes me wonder even more exactly what astaxanthin is doing.
I think they are both potentially useful. They both stimulate AMPK. It’s hard to say which is better at this time.
Do you think that drinking Aronia berry juice would be a thing to try? It’s claimed to be very high in polyphenols and resveratrol, more than Elderberries etc.
There do seem to be some health benefits to polyphenols in general. IDK specifically about Aronia berries, but they might be great!
Isn’t resveratrol a potent estrogenic? I was reading up on some forums which mentioned that resveratrol is a phytoestrogen and stillbenoid and upregulating SIRT1 might not be a good idea?
https://raypeatforum.com/community/threads/sirt1-is-a-key-promoter-of-leukemia-and-other-blood-cancers.29394/#post-437504
https://raypeatforum.com/community/threads/dont-be-conned-by-the-resveratrol-scam.1316/
Sometimes things that are beneficial in the normal state become “bad” in the cancerous state. Certainly the obese have a low NAD+/NADH ratio and low SIRT levels. In that context, thinking about strategies to reverse that is important. I am not wedded to resveratrol, although it does seem to stimulate SIRT1 (somehow).
What up with green coffee extracts, wasn’t that hyped as a belly fat shrinker years back? Polyphenol content?
Brad, do you have any ideas for my 30 yr old daughter with lipedema? She has 1 5 yr old and can’t have more due to the hormone effect on her condition. Thank you. Jan
Outside of my area of expertise. Are you familiar with Siobhans Huggins?
https://lipedema-simplified.org/flash_briefings/episode-395/
https://www.youtube.com/watch?v=7-YKSZ6U71w
I have been taking Resveratrol for various years now in the form of Japanese Knotweed root. It’s typically 50% Trans-Resveratrol which is supposedly the kind we want. So two grams of root powder (4 big caps) gives me one gram.
I started on it due to chronic Lyme disease and the recommendation by my doctor-aunt to read Stephan Harold Buhner, who, in his book “Healing Lyme” puts it at the top of the list. He’s a highly scientific herbalist and his treatises on the herbs are very detailed.
Then, later, in the anti-ageing world, I learned that David Sinclair has popularized it as well in his research on Sirtuans.
I do not believe that 5 years of taking a gram daily has had any negative effects as far as I can tell. But it has helped tremendously with chronic Lyme.
But Ray Peat, well, if you’re a Ray Peat follower, then you will have a different world view based on his various theories.
“When the electrons flow through the chain, they are eventually combined with O2 to make water.”
Surely you mean protons (H+) are combined with oxygen to make water