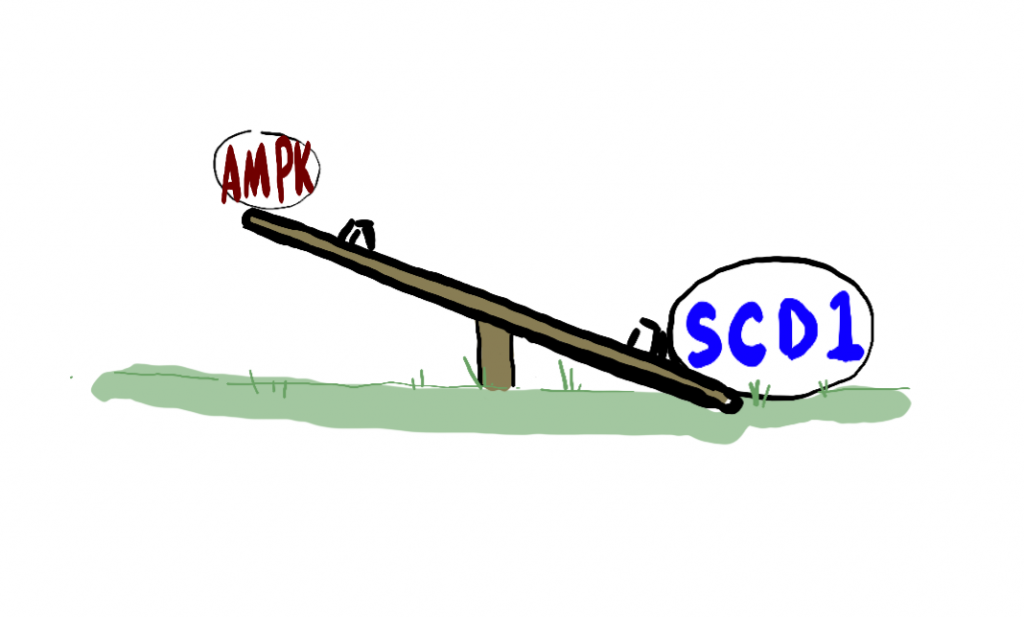
So like I was saying, biology is weird. AMPK and SCD1 are a literal teeter totter.
When SCD1 goes up, AMPK goes down. When AMPK goes up, SCD1 goes down.
This matters because, as I pointed out in the last post, AMPK can help to keep us out of torpor.
In hibernating animals and obese humans, SCD1 wins the teeter totter war. (The analogy is kind of backwards because to win at teeter totter you need to be low to the ground. Think of winning at teeter totter because you have “more of”.)
SCD1 Turns Off AMPK
In this study1, there were mice who had their SCD1 gene genetically removed and a there were mice who produced extra SCD1 in their muscle tissues. Mice who can’t make SCD1 are protected against obesity and mice who make extra SCD1 are very obesity prone.
The mice were put on either a normal “chow” mouse diet or a high fat, high sugar diet known to make mice fat. Look what happens to their AMPK levels. AMPK isn’t activated until it gets “phosphorylated”. That’s the little p. So “pAMPK” is the one we care about.
This image is a “Western Blot”. Don’t worry about the details other than that a darker band means more. There are two individual mice under each scenario. So the first two columns of the first block are normal mice on a normal mouse diet. They have a little bit of pAMPK. The next two columns are two different normal mice on a high fat diet. They make a little less pAMPK. The fifth and sixth columns are two mice on a normal diet who have had their SCD1 gene removed. They make a LOT of pAMPK. When SCD1 goes down, pAMPK goes up. Columns 7 and 8 show two mice who can’t make SCD1 on a high fat, high sugar diet. They have less pAMPK than the mice in columns 5 and 6, but still way more than mice who can make SCD1.
ACC is a target protein of pAMPK. It is responsible for lipogenesis: fat making. when it gets phosphorylated it is turned OFF. So the mice in columns 5 and 6, with the most pAMPK, have turned OFF a lot of the enzymes involved in fat making.
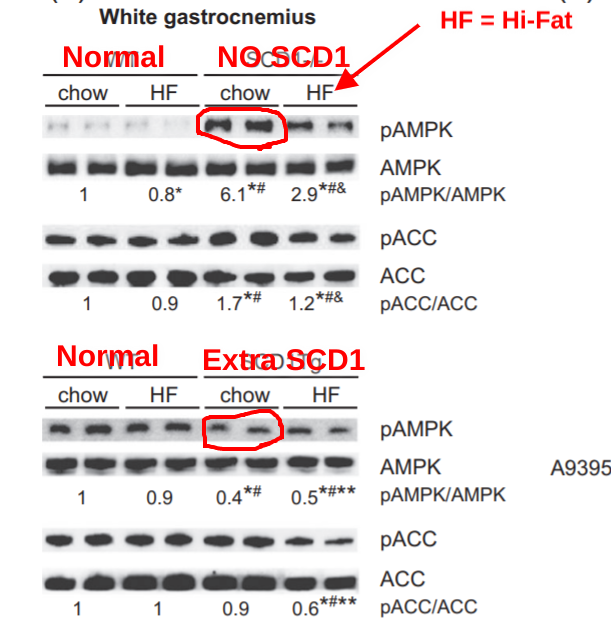
The second block shows mice who make EXTRA SCD1 in their muscle tissues. You can’t compare the darkness of the bands in the second block to the ones in the first block, the exposure time is longer. The first four columns are the same as in the first block – normal mice on a normal or high fat diet. Columns 5 and 6 are again where things get interesting. In the mice making EXTRA SCD1, pAMPK levels go DOWN 60%. This is true whether the mice are on a normal or high fat diet. But the mice with extra SCD1 on a high fat, high sugar diet have the least pAMPK activity as measured by their lack of pACC. They can’t turn lipogensis off.
AMPK Turns Off SCD1
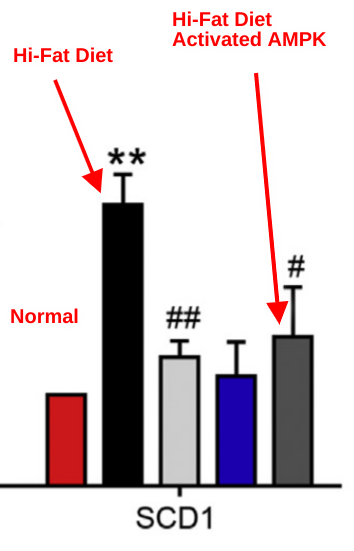
This study2 also used a high-fat diet to fatten up mice. The high fat diet caused a huge increase in SCD1 levels and the mice got fat. This fattening was preventing by berberine, which activates AMPK. Another group of mice were given a pharamcological activator of AMPK. Mice on a high fat diet given either berberine (gray bar) of the AMPK activator (labeled) had their SCD1 levels returned to almost baseline. When pAMPK goes UP, SCD1 goes DOWN.
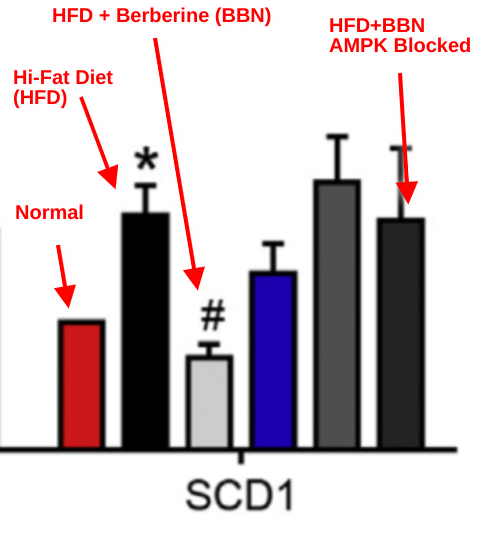
Then they took it one step further. In the next graph the red bar is a mouse on a normal diet. The black bar is a mouse on a high fat diet – SCD1 goes UP. They light gray bar is a mouse on a high fat diet supplemented with the AMPK activator berberine. AMPK goes up and SCD1 goes down. The very dark gray bar all the way on the right is a mouse on a hi-fat diet with berberine fed a pharmaceutical that blocks the activation of AMPK. SCD1 goes UP because AMPK activity went DOWN.
AMPK in Humans
This is relevant in human obesity. One of the many things that can activate AMPK is adiponectin, which rises after a meal. Except that a moderate dose of adiponectin fails to stimulate AMPK in the muscle tissue of obese humans.3
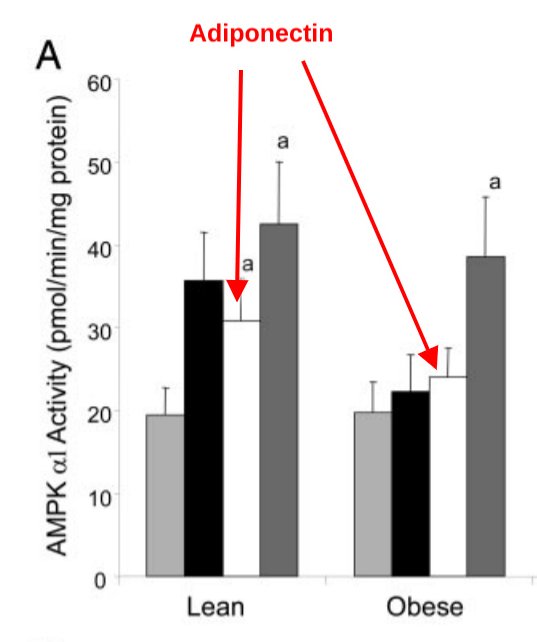
Why is this? The muscle tissue of obese humans has more SCD1 than in lean humans. When SCD1 goes up, pAMPK goes down.
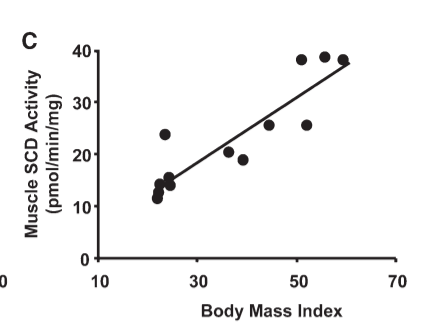
We’ve already seen that the muscle tissue of obese humans has a lower rate of fat burning than the muscle tissue of lean humans and that increasing the amount of SCD1 in the muscle tissue of lean humans causes it to behave like the muscle tissue of obese humans: it burns less fat.
pAMPK increases the amount of something called CPT1, which controls how fast fat can be shuttled into the mitochondria to be burned. So when you increase SCD1, pAMPK goes down, CPT1 goes down, fat is burned slower.
But Why?
Why would we evolve to have two genes that are critical for controlling metabolic rate, each of which suppresses the other? No human would design a system like this.
The answer is that this creates a type of meta-stable binary system. The normal state of mammals is to have relatively low levels of SCD1 which allows them to quickly switch into fat burning mode when insulin levels are low – two hours or so after a meal. This happens because AMPK is quickly activated into pAMPK. pAMPK will suppress SCD1 expression and ensure that SCD1 levels remain low. In this state, mammals burn fat efficiently and remain lean.
In response to diets high in PUFA and/or sugar, mammals upregulate SCD1. In this alternate state, mammals do not switch into fat burning mode when insulin drops after a meal. They are in fat storing mode. Fat storing mode is critical if one is trying to fatten up for winter. Once SCD1 is upregulated the mammal stays in fat storing mode because SCD1 inhibits the activation of AMPK.
It’s like a teeter totter and you don’t want SCD1 to win.
In the next post I’ll look at the underlying mechanisms that control the relationship between AMPK and SCD1. There will also be a some posts about ways to increase AMPK activation but you want to do some research, I’ll give you some hints: Vitamin D, Vitamin K2, berberine, metformin, resveratrol.
You can also block SCD1 activity with Sterculia Oil.
- 1.Dziewulska A, Dobosz AM, Dobrzyn A, et al. SCD1 regulates the AMPK/SIRT1 pathway and histone acetylation through changes in adenine nucleotide metabolism in skeletal muscle. J Cell Physiol. Published online June 26, 2019:1129-1140. doi:10.1002/jcp.29026
- 2.Zhu X, Bian H, Wang L, et al. Berberine attenuates nonalcoholic hepatic steatosis through the AMPK-SREBP-1c-SCD1 pathway. Free Radical Biology and Medicine. Published online September 2019:192-204. doi:10.1016/j.freeradbiomed.2019.06.019
- 3.Chen MB, McAinch AJ, Macaulay SL, et al. Impaired Activation of AMP-Kinase and Fatty Acid Oxidation by Globular Adiponectin in Cultured Human Skeletal Muscle of Obese Type 2 Diabetics. The Journal of Clinical Endocrinology & Metabolism. Published online June 2005:3665-3672. doi:10.1210/jc.2004-1980

Good stuff! You have probably seen all my linkspam on twitter but I have noticed how a lot of those antioxidants found in various teas & herbs (& plant pigments) activate AMPK according to studies. Really seems like we have a seasonal switch here. Harvest-time torpor (linoleic acid, sweet fruits) and spring-time anti-torpor (Flowers, pigmented berries, teas). It’s elegant if you ask me.
Also noticed the vitamin D reference. Another nutrient that’s lacking a bit in the wintertime – but comes about when weather gets warm, in the spring… from our own skin & sun exposure! Vitamin K less clear but a lot of leafy veggies have it.
PS- Another topic of curiosity- When we’re using AMPK agonists to try and disable torpor, does calorie expenditure increase automatically, or is it proportional to muscle usage? (i.e. does the “real” weight loss happen via uncoupling in proportion to acute muscle calorie usage)
Might be skipping ahead on the series with this question but just planting it here as a thought for future posts.
Vit K is in Leafy veg. Vit K2 not so much. K in the gut is converted to K2 by bacteria mainly in herbivores. the bit converted in our gut is in distal colon and virtually unabsorbed. Most of the popular description of K2 pertains to carboxylation of GLA protein and its impact on calcium partitioning. I expect what Brad is referring to is a lesser known property of K2 where it functions as an alternate electron transport chain. A very cool study at N Texas U showed a 12% increase in cardiac output after a short period of K2 supplementation. I wait with anticipation his observations.
One thing that happens with mice is that in response to leptin – which activates AMPK – they spontaneously get up and walk around more often.
So, since fasting activates and uses the AMPK pathway on steroids, we are safe to assume that fasting acts to both detox PUFA (as they are burned preferentially), AND inhibits SCD1. Am I just reading this wrong, or has nature given us the answer for torpor all along?
Makes sense, that is what torpor is for anyway- preparing for fasting when food availability will be low. I think Brad’s take is this just isn’t ideal for modern Human society and perhaps there’s a chemical/nutraceutical approach.
Fasting works, but you have to fast until all or at least most of the PUFA is gone. We aren’t hibernators so that would be rough.
Actually, SCD1 is increased in response to fasting, at least in lean humans. This way, the next time you eat, you will store more calories. I’m not sure if this holds true in obesity.
This is interesting, I wonder what the pathways look like.
Anecdotally I’ve found with short term IF when I refeed I feel tired after, especially with lots of carbs (starch). This morning I was sipping hibiscus for hours before refeeding and I was still pretty shocked how tired I got when I ate lunch (skipped breakfast)
Hello Brad!!
you know i have been trying to lose weight and get well for about 25 years, and always got the same two answers on youtube, no fat veganism, and keto/carnivore… i can’t stick to either of these, they are torture! then.. i said a prayer and asked for a diet i can actually stay on, and would work, and lo and behold there you were!! i feel like i fell into an alternate universe! i’ve never seen you before!
I’ve been trying your diet with good hamburger, rice, steamed veggies with a coffee with butter and cacao butter in it
so far have not lost weight, but i had to, just had to try some basic cookies with just flour salt sugar and butter… gained 3 lbs, sugar bad
anyway, i wanted to talk to you about torpor, i know this is what’s wrong with me, don’t ask lol but i have never heard how to get out of it, i ordered your butter oil stuff, but can’t afford the other until the end of the month.
i have a story to tell you, i can’t lose weight without a LOT of exercise, i can jog for 6 months without losing an ounce, then suddenly something kicks in and i drop weight so fast i don’t notice it, people have to point it out to me!
and my whole personality changes, the depression lifts, i become super ambitious etc etc
can you tell me, since i can’t run anymore coz of body pain, do you know what it is that ‘switched on’ in me, and if there’s some way i could simulate it?
thanks so much for your consideration, i know this is a weird question! xoxoxoxoxox
Well, that is a very interesting story. I can’t say for sure what changed in you! Perhaps AMPK was activated. But why? Citrus? SUmmer? Vitamin D?
Brad!!
I forgot to tell you!!
when I gain weight, like when i stop exercising, i reach a point where i can ACTUALLY FEEL IT, and i feel ‘water logged’ my body feels like all the cells are getting bloated with water!!! and i can gain 40 lbs over a 2 week span without even noticeably changing the way I eat!!! and my mother gained weight in chunks the way I did as well, and neither of us could really lose weight on ANY diet!! my mother died of cancer still fat!!
So as I listen to you, i realize that my ‘switch’ turns on fast and turns off fast and it has to do with my fat becoming liquid or not…
Seriously, this just hit me!!
I tried another diet recently and it had a cheat day once a week, by Tim Ferris, and I did it for two weeks and gained 7 lbs on each cheat day, and that 14 lbs is starting to come off sloooowly with your diet! maybe I will be like Emmy and lose not much at first and then it will kick in?
Anyway, this must have to do with a cascading effect that works very quickly, changing my entire composition from saturated to PUFAs, but why do i gain more so fast, and lose weight so fast? why so much, so FAST?
Perhaps you should study me! Come to Canada lol
switch, turns my fat to PUFAs, gain a ton fast
switch, turns my fat to saturated, lose a ton fast
doesn’t that pique your curiosity?
I’m watching some woman on Bulletproof radio talk about vegetable oils, she doesn’t know as much as you do… but I think I have watched all the vids you have out there 🙂
cheers
Elaine xox
Interesting. Of course there can be multiple factors at work here. Glycogen repletion, AKA what many people call “water weight”. Also, of course, switching on lipogenic genes and therefore MUFA.
Quicksilver Scientific sells a product called AMPK Activator. Its liposomal format so it flips the switch quick, like 90mins. Its ingredients are familiar, berberine, quercetin, resveratrol, milk thistle, cinnamon bark. There are lots of non liposomal ampk activator products on the market……..
Let’s not forget that exercise is a very strong activator of AMPK.
Should add that a few more substances activate AMPK…genistein, EGCG and capsaicin.
https://pubmed.ncbi.nlm.nih.gov/16236247/
Also seen coffee can do it (chlorogenic acids)- https://pubmed.ncbi.nlm.nih.gov/23416115/
Hibiscus tea seems to have a bunch of plant polyphenols that do it- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5579700/
Brad mentioned Amaro bitter liquers – https://twitter.com/fire_bottle/status/1437791792984989698
There are a whole host of things that increase AMPK. Almost too many to list, so I’m focusing on specific cases that point out fundamental aspects of the system.
Interesting tidbit from Peter’s interview on Peak Human podcast- Peter mentioned that he suspects on some level, all the metabolic signaling inside the cells ends up boiling down to ROS in one way or another. As you mentioned before when folks think “antioxidants”, think “ROS brokers” or somesuch.
Setup for this comment: https://high-fat-nutrition.blogspot.com/2021/09/modelling-energy-intake-2-corn-oil.html
This brings me to another question- ROS from mitochondria from linoleic acid. IIRC, part of why linoleic acid lowers ROS is not so much the lower FADH2/NADH ratio, but the fact that PUFAs often trigger uncoupling proteins – in extreme examples Peter’s cited studies where >40% linoleic acid actually prevents obesity due to the prodigious use of uncoupling (and a really sweaty organism)-
The effect of uncoupling is to lower the “voltage”, as I like to think of it, or “delta psi” as Peter describes it. When the voltage is lower due to high uncoupling, less ROS is produced by a given FADH2/NADH ratio of fuel fats. So having a moderate amount of linoleic acid – and a commensurate amount of UNCOUPLING – effectively trashes the ability for ANY fatty acid to produce high ROS, right?
And so what these “antioxidants” are doing, is adding exogenous ROS to our cells, to cover for linoleic acid’s uncoupling effect?
I would say that in low amounts, PUFA limits the ability of other fats due to it’s effect on the FADH2:NADH ratio. At very high amounts, PUFA breaks down into 4-HNE, etc, which cause uncoupling and weight loss. I would argue that uncoupling and thermogenesis are what we want from a weight loss point of view – the easy burning of calories due to “inneficient” metabolism, but that we want to get there through mitochondrially produced ROS via high FADH2:NADH ratio rather than general oxidative stress via PUFA/4-HNE.
Just an anecdote…have been trying to increase my BMR and using a simple thermometer and basal readings in the morning to monitor or throughout the day.
Initially , the strategy began with severely limiting PUFA (ie N6 mostly) and increasing SFA. I eventually then added berberine and sterculic oil and got a response but stopped both of the supplements because of digestive issues (maybe not related but stopped anyway). In the meantime I took several Omega quant tests (so this was going on over six months +)which showed lowering of LA and DI18 and DI1600 and EPA/DHA ratio increased (no supplementation O3). Significantly. However, temperatures did not rise much beyond 98F max in mid day. Basal around 97F but up from mid 96’s. Exercise , but Maffetone type HRZ2 intensity. No HIIT or weight training.
More recently, developed some increasing basal temperatures like 97.4-5F and mid day or early evening 98+. But to address the comment about sweating…..in past did not sweat hardly at all. Now sweaty just thinking about exercise…and salty sweaty on my shirts and hats. Something is happening but I need to find access to a calorimeter. Only supplement is a vitamin/mineral cap and drinking now LMNT electrolyte solution. Na, P, Mg. My guess is that for whatever reason my mitos started kicking in and system generating excess electrons which ended up in uncoupling to protect the ER stress reaction. Uncoupling is good (see Ray Peat too)
Stable weight or very modest weight loss. Probably eat too many croissants. Wife makes homemade. How can I not eat them? At least I exchanged the chocolate ones to gruyere and plain butter croissants!
Very much considering a low supplementation with Omega 3’s. …and see if that moves the OQ test needle any further. Not quite into their optimum range but started way out of it in the beginning. Still can track DI and AA.
Very interesting feedback!