Introduction
My thesis in this series is that humans get fat NOT from sloth and gluttony, human characteristics wrapped in morality and asceticism. They in fact get fat from torpor, an underlying metabolic change triggered by consuming linoleic acid. In my previous article I introduced the concepts of this series as well as the idea that sedentary Americans ate more in the 1930s than they do now. Yet obesity wasn’t a problem then and it is now.
The authors of the paper I will focus on in this post show that much like in hibernating animals humans store fat BECAUSE OF increased amounts of an enzyme called SCD1. SCD1 causes torpor in mammals, a metabolic state that is a pre-requisite for hibernation and which favors fat storing over fat burning.
The authors of this paper culture muscle tissue from obese and lean Americans. They show that muscle tissue from lean Americans burns more fat and stores less fat compared to the muscle tissue from obese Americans. They further show that increasing SCD1 in the muscle tissue from lean Americans causes that muscle tissue to burn less fat and store more fat.
Thought question: Does increasing the level of SCD1 in isolated muscle cells of lean Americans cause those muscle cells to become lazy? Are the muscle cells overeating because of their obesogenic food environment? OR is it more likely that metabolic changes are the cause of obesity? Is it more likely that cells can CHOOSE whether to burn or store fat? As the authors of this paper write, could it be that obesity is not a moral failing but a disease of “abnormal fatty acid partitioning”?
Hibernating hamsters increase SCD1 to store fat for winter
As a Syrian hamster gets ready to hibernate, it switches into an alternate metabolic state called torpor. It does this in large part by increasing the expression of a gene called SCD1,1 whose function is to unsaturate the hamster’s body fat.
When fat is very unsaturated, animals can resist the process of thermogenesis (heat making) which is driven by a hormone called leptin which is produced by fat cells. Leptin sensitivity leads to fat burning and the goal of torpor is to not burn fat.
Modern humans increase SCD1 to store fat for nothing
This is the paper2 proving that SCD1 isn’t just correlated with torpor and fat storage, it actually CAUSES it.
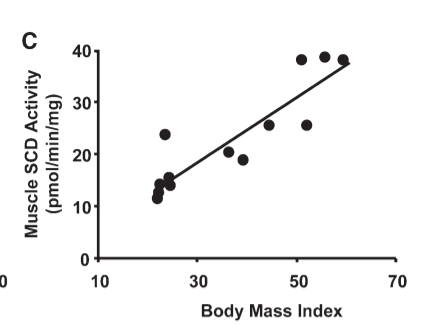
The authors show there is a very tight correlation between the amount of SCD1 activity in human muscle tissue and BMI. Very obese humans make a lot of SCD1. The more obese you are, the more SCD1 you make. Just like in torpid animals there is a high correlation between SCD1 levels and body fat levels.
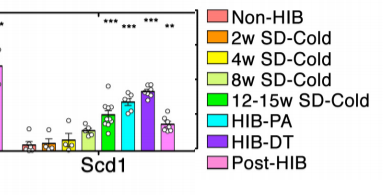
They also looked at which genes are increased in the muscle tissues of obese humans. It turns out the answer is “mostly SCD1”.
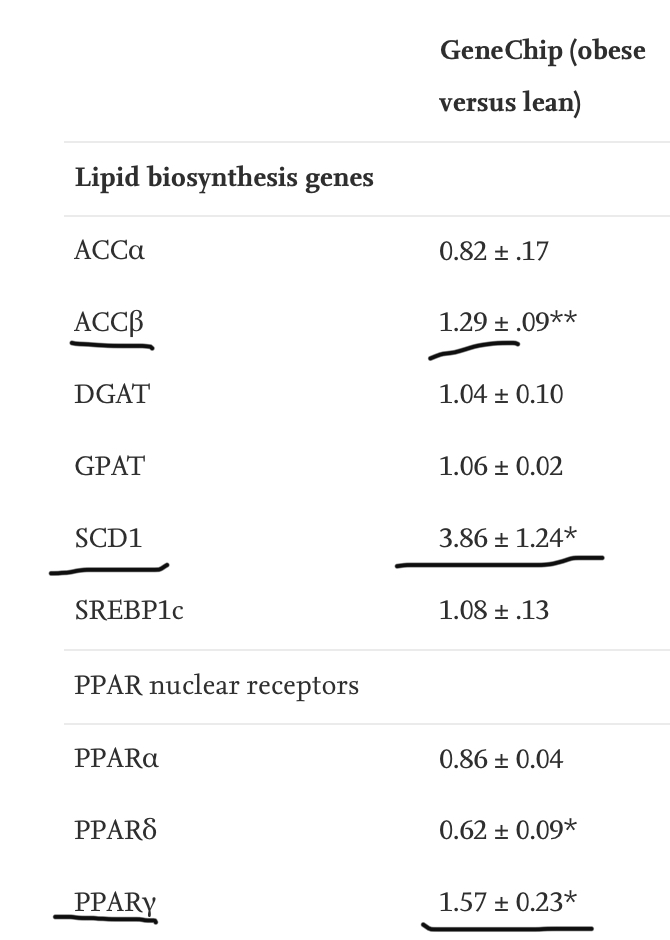
Of all the genes involved in fat synthesis and storage, SCD1 was the single gene that was the most increased in obese humans – by a factor of 4. SCD1’s co-conspirators PPAR gamma and ACC beta were also increased in the obese, but that’s a story for another day.
What the authors show across the board is a correlation between SCD1, desaturase index and obesity. Of course, correlation isn’t causation. Happily, this research group is very clever. They took tissue cultures from the muscles of lean and obese people. It turns out that the muscle cells of obese people compared to those of lean people:
- Burned less fat
- Made more fat
- Stored more fat
- Had 6 times as much SCD1 protein
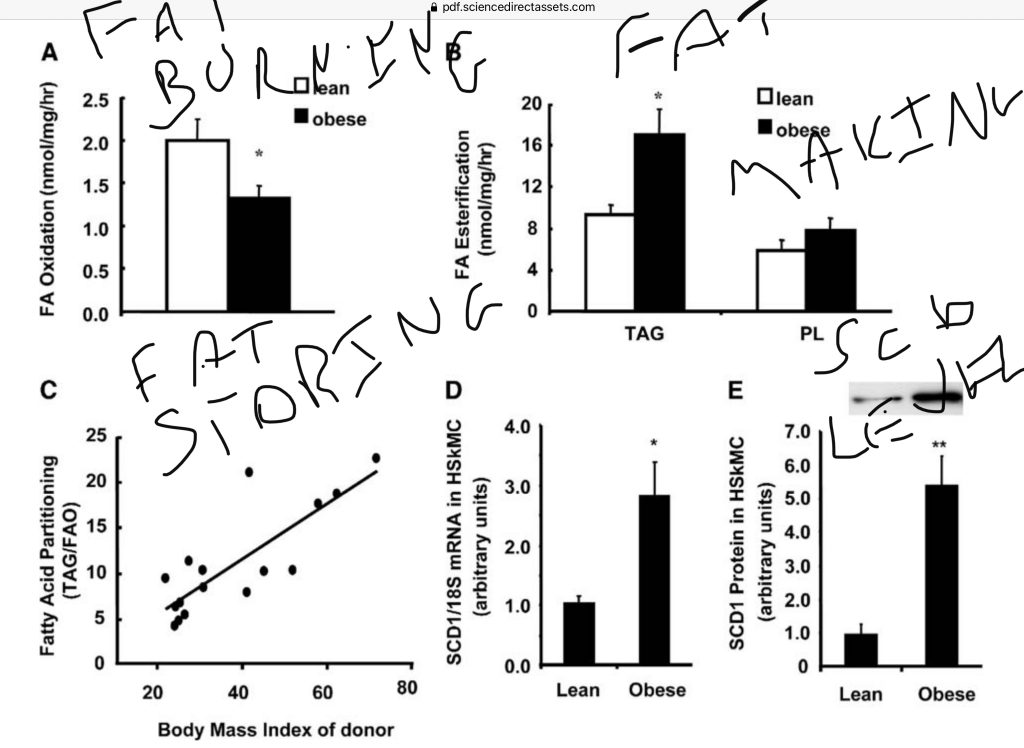
So we can say that there is a strong correlation between SCD1 levels in skeletal muscle, the rate at which skeletal muscle makes and stores fat and an inverse correlation between skeletal muscle SCD1 levels and fat burning. Correlation still doesn’t imply causation. To test whether SCD1 CAUSED these metabolic differences, the authors overexpressed the SCD1 protein in the muscle tissues of lean humans. Guess what happened?
Muscle cells of lean humans whose SCD1 levels were doubled burned less fat, made more fat and stored more fat.
SCD1 was sufficient to make the muscle cells of lean people behave just like those of obese people!!
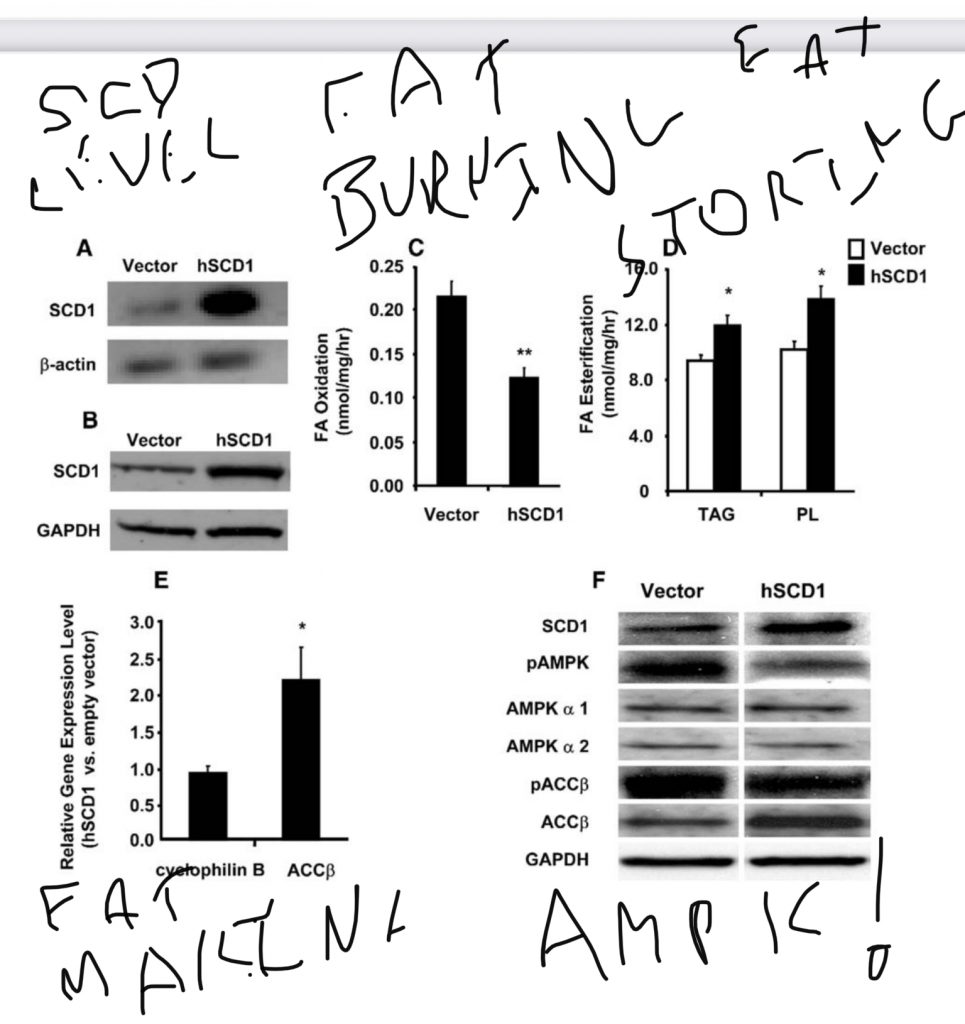
This allows us to say that SCD1 isn’t just correlated with low levels of fat burning and high levels of fat storing, we can say that it CAUSES them. We can say that it causes them in the muscle cells of actual live normal American humans.
A possible mechanism by which SCD1 exerts its effects is by decreasing the levels of phosphorylated AMP Kinase (pAMPK), an enzyme which is a “Master regulator of metabolism”. Guess one of the ways leptin exerts its fat burning effects? By increasing phosphorylated AMPK.3 Leptin and SCD1 are eternally at odds.
How to Lower SCD1
If you follow this blog, you’ll know that I am collecting a list of things that lower the level of or block the activity of SCD1. SO far, to date, we have these things which lower the expression levels:
There is also an oil that blocks the activity of the enzyme that I sell. You can find my recipe for escaping torpor. Come back, the recipe will be evolving as we learn more!
- 1.Chayama Y, Ando L, Sato Y, et al. Molecular Basis of White Adipose Tissue Remodeling That Precedes and Coincides With Hibernation in the Syrian Hamster, a Food-Storing Hibernator. Front Physiol. Published online January 28, 2019. doi:10.3389/fphys.2018.01973
- 2.Hulver MW, Berggren JR, Carper MJ, et al. Elevated stearoyl-CoA desaturase-1 expression in skeletal muscle contributes to abnormal fatty acid partitioning in obese humans. Cell Metabolism. Published online October 2005:251-261. doi:10.1016/j.cmet.2005.09.002
- 3.Minokoshi Y, Kim Y-B, Peroni OD, et al. Leptin stimulates fatty-acid oxidation by activating AMP-activated protein kinase. Nature. Published online January 2002:339-343. doi:10.1038/415339a

Brad – back in part 2 of your SCD Theory articles (November 16, 2020) you referenced a Fructose rodent study. It wasn’t cited at the end of that article, but I believe it’s this one: https://www.liebertpub.com/doi/10.1089/jmf.2019.0177. In the article, the authors reference a daily intragastric dose of 0.06g Sterculic Oil per day. If we assume the rodents had an average bodyweight of 250g over the 8 week study (they started around 110g and approach 390g at the end), that suggests something like 0.025% SO:Bodyweight ratio. For a 200lb human (i.e 93kg) at the same ratio I make that 22,000mg per day, and not the 2,200mg per day which is the dose you’re suggesting. Apologies if there’s something wrong with my mathematics..grateful for your confirmation!
I’m recommending less than those seen in mouse studies. Remember that humans have a much lower metabolic rate per lb than do mice. That article says the dose was calculated for 1% of sterculia oil in the diet. I assume that means 1% by dry weight (mouse chow is dry). So a human consuming 2400 calories, with 40% of it as fat, would be consuming roughly 6 calories per gram, which is to say 400 grams per day of dry weight. If 1200 mg of this is Sterculia Oil, that’s around 0.3% of diet. That’s around a third of what the mice got, maybe a fourth considering fiber, etc. So I’m suggesting less, but not as much less as you’re suggesting I am.
This would be the optimum time to insert a video of Chevy Chase saying “Not that I have a Log. At least not in the sense that you think that I said that I did” from Christmas Vacation. I don’t think I can do that in the comments.
Have you lost weight using this recipe?
I plan to try it as I already take metformin and berberine for Type II diabetes. Have been stuck at fixed weight for 4 years even while being on keto diet with low level ketones. Also take hypo thyroid meds since 13 now 70.
Paul, for me, the berberine does seem to have some effect, mainly in some lack of hunger and also eating smaller meals (sometimes, anyway). I cannot tell the sterculia has any effect, although I find it hard to take. Since I don’t eat breakfast, and taking berberine on an empty stomach made me starving, I take 1 gram berberine with my first meal. I also drink 1-2 cups of Pu-erh tea in the morning before my first meal.
I take the sterculia oil later, at my second meal. But it seems to have negative digestive effects. I try to take it as many days in a row as I can, but that’s only usually Monday-Thursday, then I give up. I can’t tell any effects, such as higher body temp. Or really anything at all. But I eat, then go to bed no long thereafter. (This is not ideal, but this is my schedule.)
In my experience it’s the stearic acid that will make your body temperature rise. If I understand correctly, supressing SCD1 will stop it from desaturising your fat. But that does not mean your already unsaturated fat will burn off.
It’s worth giving a try.
Tom, this is my experience. My bodyfat is relatively unsaturated (my desaturase index is 1.9) and I’ve been supplementing berberine and sterculia oil for a month with no material impact to body temperature.
I personally see a slow rise in temperature with sterculia oil. This takes several weeks at least. Over time your body fat will become more saturated. It’s unclear how long the changeover will take in an adult human.
Brad
The way sterculia oil has work for me has been to eat more saturated fat (that I also buy here) avoiding polyunsaturated fat at all cost, and not overeating. So during the weekend I tend to overeat and this has trapped me in a cycle of losing and gaining a few pounds each week. After a month or so after taking Sterculia oil, I began to noticed my capacity to gain weight has been reduced. First a few grams and now almost a pound, so now is easier to lose weight just by a hair. So if I was gaining 8 lbs during the weekend now I just gain like 7 by intermittent fasting I lose 8… But it took a while, sadly when I say I over eat I mean in a dittion to my carnivore meal I will have some 1 or two pieces of starchy cheese bread and few alcoholic drinks, may eat outside some Vietnamese food, just 2 meals a day… Clearly my fat cells are insulin sensitive as hell.
I expect the stercuia oil to have long term effects. Not short term ones. After five months at the 600-1800 mg dose I am seeing body temperature increase.
I think I am starting to. I also think it will take a long time to convert over body fat – perhaps a year. But I think I’m headed in the right direction.
Awesome thanks Brad. Can you please share an update on how you are taking the supps? At one I believe you may have been taking them all at once at night; and then I might have seen somewhere else that you were splitting up? Would love to know what you think works best. Thanks for the amazing info!
Yeah. Umm… I should probably be more disciplined. I was taking berberine separate from my main meal on the assumption that berberine is short lived and I didn’t want the berberine to mess with ROS production during the post-prandial stage. But then I was just forgetting to take it at all. Now I’ve been doing it with meals, but only every other day or so. I am taking astaxanthin daily with meals.
I’m also beginning to experiment with resveratrol and hesperidin… NOT because they are anti-oxidants but because it seems they down-regulate certain micro-RNAs (miRNA107, for instance) who are in turn down-regulating rates of fat oxidation.
Brad
On the off-chance you aren’t aware of these:
https://www.youtube.com/watch?v=6Ik0IFaa2Oc (and there is a part 2 as well)
Shots fired!
I haven’t given them a close listening-to yet, so I’m not sure how solid their critiques are.
I’ve listened to both of them. I thought the first one was pretty good. The second one missed the mark pretty badly. I think they missed the core of my argument. Having said that, it’s been long enough since I listened that I can’t remember exactly where I agreed or disagreed with them.
Brad
Why is Astaxanthin not on list?
Wow good call on this. After reading your comment I started looking more into astaxanthin. I knew about a bunch of health benefits but didn’t realize it down regulates SCD1 and upregulates PPAR alpha. Really interesting stuff.
I started seeing my basal temp rise after starting astaxanthin 12mg bid. Sterculia oil gave me digestive issues as Berberine. However, is it the case that eating lots of sfa’s will end up in muscle fat and result more stored fat even if PPARs are elevated but scd1 not inhibited?
The scenario is possible. In theory, when PPAR alpha is highly activated it should help suppress SCD1, but with torpid body fat, it’s unclear whether that will work. The mechanism that suppresses SCD1 may well be mitochondrial ROS production, which is hard with unsaturated fat.
Brad
It should be. I’m lazy.
DHEA is also a PPAR-alpha activator
https://onlinelibrary.wiley.com/doi/full/10.1111/j.1474-9726.2012.00852.x
Just spent a couple of days catching up on this site and going down a few biochemical rabbit holes. Such interesting information here. Lost a fair amount of fat on keto last year, had been maintaining over the holidays and early spring as I moved into a more relaxed diet. Within the past couple of weeks I have been focusing on keeping PUFA low, adding stearic acid through Brad’s SA butteroil and cacao butter, adding in a few starches. I am also using berberine 500mg twice daily and the sterculia oil with the morning berberine dose. Haven’t been on a scale but pants are a bit looser and my hands and feet are warm, where they used to be cold much of the day and night.
In addition to this site, I have been reading various stuff including Ray Peat, and I have been including small quantities of fruit and juice, with a bit of real-food-only ice cream or chocolate here and there. I am 49 with still a fair amount to lose, but I feel like adding back in some carbs and paying
attention to my metabolism is important. So far, my subjective assessment has been that the research here is working for me.
Excellent, Jen! Thanks for the feedback. Good luck!
Brad
In one of the blog’s post about stearic acid, you wrote :
“since I published the article, some experienced quick weight loss and waistline reduction, but others had quick weight gain and a waistline increase.”
I do wish we could know more about those who had “quick weight gain and a waistline increase” and why the result of the diet was seemingly the strict opposite of expected results.
Has your comprehension evolved on this topic ?
In that comment I’m referring to combining starch and saturated fat. For some this has caused weight loss, for others it has produced gain. I think this is likely tied into how torpid you are. I consider this a separate issue from stearic acid.
Brad