AKA The Ros Theory of Obesity Part2, Part 2
Introduction
In part 1, I explained that SCD1 is up-regulated in things that are fat and that in fact up-regulating SCD1 in something is sufficient to make it store fat. Insulin is trying to upregulate SCD1 while leptin down-regulates it. The mechanism by which leptin down-regulates SCD1 is by increasing fat oxidation in the mitochondria which drives ROS production, which leads to uncoupling proteins being expressed which allows you to burn off excess calories as heat. We also saw that weight loss trials using an injected form of leptins failed to cause weight loss in Americans.
In part 2, we’re going to take closer look at how SCD1 is regulated. A small amount of mitochondrial ROS actually increases SCD1 because a little ROS is the signal that glucose is being burned and when fat is made from glucose it has to be somewhat desaturated to maintain membrane fluidity. If your body fat is high enough in PUFA, the small amount of ROS produced by your mitochondria looks to the cell like glucose oxidation, so SCD1 gets up-regulated even while fat is burned! This leads to less saturation, which drives less ROS, which upregulates SCD1 even further – a vicious cycle of unsaturation leading to the obese physioogy.
Then we’re going to look at how you can test your SCD1 levels, a variety of supplements you can use to lower it and I’ll introduce a natural inhibitor of it.
Part 3 looks at how dietary monounsaturated fat (MUFA) fits in.
A Little ROS Increases SCD1. A Lot of ROS Decreases it.
Buried on page 11 of this great paper, is a graph that shows that a small burst of ROS dramatically increases SCD1 expression.1 It takes a LARGE increase in ROS to suppress SCD1 production.
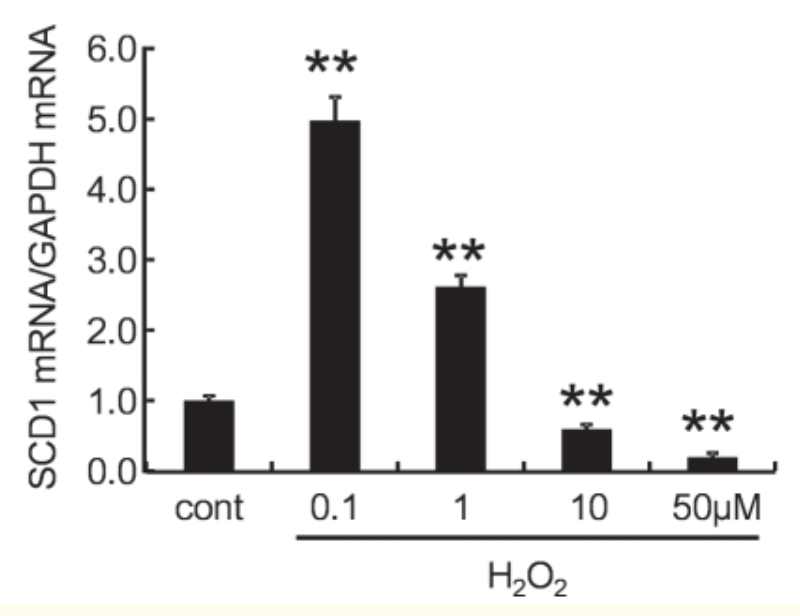
This is biologically appropriate. When glucose is being burned, a small amount of ROS is produced from cytosolic NADH produced during glycolysis.2 A person living on a high starch diet – white rice – has to produce some SCD1. They need body fat for building cell membranes, fatty tissues, etc. They have to make this from starch and a certain amount of MUFA is required in that case for proper membrane fluidity.
When sufficiently saturated stored body fat is being burned at the request of leptin, lots of ROS is produced and SCD1 is inhibited, initiating the cycle of leptin induced thermogenesis. BUT! If that stored body fat has enough PUFA in it, ROS production will be insufficient to drive enough ROS production to shut down SCD1 and in fact SCD1 will be increased!
Burning PUFA in the mitochondria looks to the rest of the cell as if the mitochondria is burning glucose. The level of ROS production is a signal from the mitochondria to the rest of the cell to tell the nucleus in which phase of the Randle cycle its in – whether it’s burning fat or glucose – so that the nucleus can respond appropriately. The cell does the appropriate thing in the context of a small increase in ROS production: it increases SCD1. Except that in this case the stored fat is already highly unsaturated. More SCD1 is produced and the fat gets even more unsaturated.
This is a positive feedback loop going in the wrong direction!! This loop is unsaturation leading to more unsaturation leading to a pathological inability to up-regulate thermogenesis in response to leptin.
The Western Diet and The Post Obese
This is a really stunning paper published in January.3 The authors used a synthetic drug called alpha-naphthoflavone (aNF) to disable the “aryl hydrocarbon receptor”. This has a number of effects but one of them is dramatically reducing the production of SCD1. aNF was able to prevent obesity in mice caused by a “Western” diet.
A Western diet, as defined by obesity researchers, is one than combines starch, sugar, lard and soybean oil. It is typically a “high fat” diet, getting 45-70% of its calories from fat. It is said to be high in saturated fat, but I differ on this point. American lard is high in both polyunsaturated and monounsaturated fat.
These are the diets used in this study as the Western and control diets:
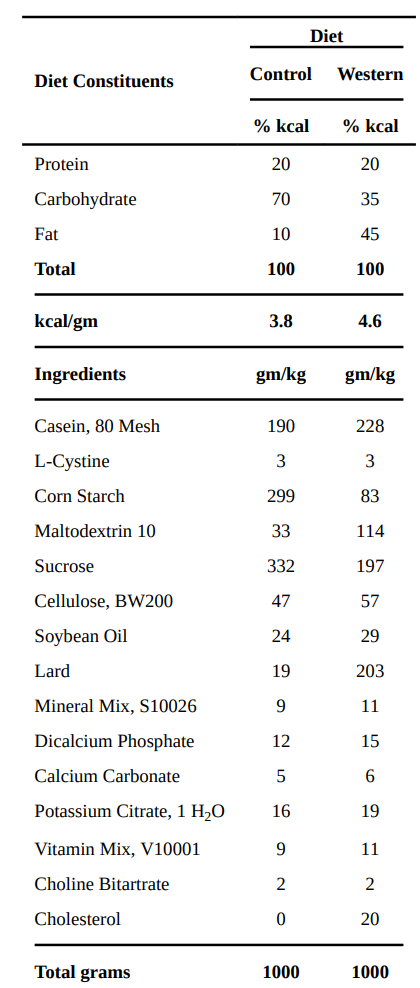
A friend just sent me samples of porkfat they had tested. The ratios of fat in it were 32% Saturated, 45% monounsaturated and 16% polyunsaturated. My guess is that this is pretty close to average for american pork. With that assumption, this diet comes out to around 9-10% of calories as PUFA, 19% of calories as MUFA and 13% of calories as Saturated Fat. It’s a very unsaturated blend! As I’ve already written, this is plenty of polyunsaturated fat to fatten up susceptible mice.
Conversely, a mixed diet of starch and highly saturated fat causes leanness in mice.
OK, let’s look at the study results.
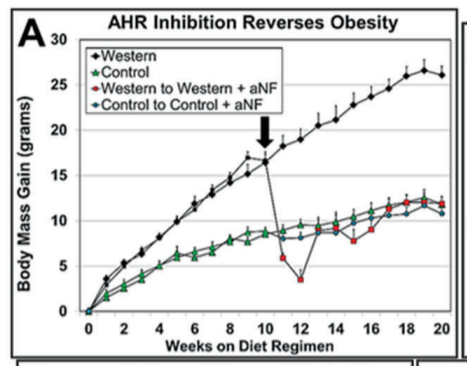
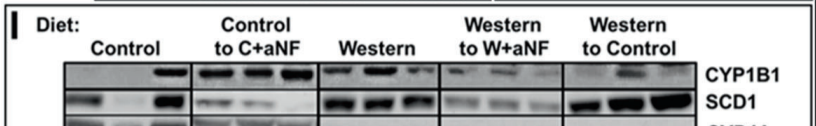
In graph A, you have two groups of mice – one is on the control low fat diet, the other is on the Western diet. The Western diet mice get fat. Then after 10 weeks, half of the mice on the Western diet are given a pharmaceutical that reduces their SCD1 production to almost nothing. Immediately they break out of the positive feedback loop and become lean! Mice have a much higher metabolic rate than humans so the change happens very quickly.
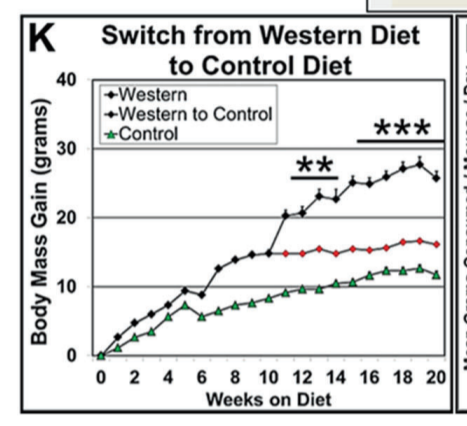
In graph K a very similar experiment is done except once the mice are fat some of them are switched back to the low fat control diet. Switching the mice over to the control diet prevents further weight gain from the Western diet but those mice never return to the lean state. They are post obese!
Their SCD1 levels remain elevated after the dietary switch. They are locked in a positive feedback loop of unsaturation leaving them with an inability to do leptin induced thermogenesis.
I Am Post Obese
I resemble the post obese mice in a lot of ways. My body temperature is low – typically around 97.2. I’ve lost a lot of weight by increasing the amount of saturated fat I eat and reducing minimizing dietary MUFA and PUFA. I have to be very vigilant, though. If I was strict I’m sure I could stearic acid macro-dose myself to a much lower weight. But with a post obese metabolism I know that as soon as my control weakens the pounds will begin to slip back on. I am certainly not rapidly returning to a lean state without concerted effort like the mice who had their SCD1 levels pharmacologically lowered did.
I decided to test the hypothesis that like the post obese mice I have an elevated expression of SCD1. The ratio of oleic acid to stearic acid in your tissues is a fairly reliable indicator of SCD1 levels. Researchers call this ratio the Desturase Index. A high ratio is an indicator of high SCD1 levels. Dietarily, I have been maximizing my stearic acid intake and minimizing my oleic acid intake for well over a year now, so my body has had plenty of stearic acid to store.
This is the test I did.
If you’re struggling to lose weight I suggest you take the same test to see where you fall.
| Oleic Acid | Stearic Acid | Desaturase Index | |
| Me | 23.2 | 10.8 | 2.1 |
| China Health Study | 14.4 | 15.5 | 0.9 |
| Nathan, a Lean American | 21.6 | 13.3 | 1.6 |
Clearly my fat cells have been busy converting all of that stearic acid I’ve been eating into oleic acid. I am trapped on the wrong side of the mountain!
The test shows the desaturase of your red blood cells, so it won’t be reflective of your stored fat but it’s an easy and relatively affordable test to do, results are fast and I think comparing yourself to myself, Nathan and the Chinese will give you a reasonable guess as to your desaturase activity. It shows a lot more info than just these two numbers. You can calculate your desaturase index by dividing your Oleic(18:1n9) by your Stearic(18:0).
One comparison in the table is to an average mostly-starch-eating rural Chinese person taken in the early 1980’s as reported in “Diet, Life-style and Mortality in China”, colloquially known as “The China Health Study”. As you can see, starch eaters have very saturated fat. I’ve seen the same thing in my pigs. A high starch diet makes their fat very firm, which is the basis of Firebrand Meats Low-PUFA pork.
The other comparison is to Nathan Owens, an American who is lean, 14-16% body fat and who follows a diet of “Carnivore, mostly Costco prime beef, some lamb, fish, eggs, cheese, and dark chocolate”. He had done the same blood test and posted his results on Twitter. As you can see, his desaturase index is a lot lower than mine, although not nearly as low as the Chinese.
Nutritional Factors affecting SCD1
SCD1 is up-regulated by dietary carbohydrate, both directly and as a response to insulin. This is biologically appropriate. A person living on starch needs to create saturated fat by De Novo Lipogenesis to create cell membranes, etc. These cell membranes require a certain amount of fluidity. Saturated fat is a solid wax at body temperature and so a certain percentage of our body fat has to be monounsaturated.
Sugar up-regulates SCD1 much more dramatically than starch. In the previous post we saw that mice on a very low fat, high sugar diet up-regulated SCD1 by almost tenfold!
Unsaturates
Full disclosure! The product links that follow are sponsored links. If you click on them and buy products it helps Fire In A Bottle bring you more of what is hopefully insight. I looked at a lot of different product lines and Life Extension and Super Smart were the ones that impressed me the most. They both have great lineups. I like the Life Extension AMPK activator that combines g pentaphyllum with hesperidin. If you combined that product with the berberine you’ll have an herbal triple threat. On the other hand, super smart has pure hesperidin, which might be interesting to try on its own since it’s the one that directly activates ERK1/2.
All unsaturated fats lower SCD1 production, polyunsaturated fat more than monounsaturated fat. This makes sense biologically. For instance, Native Americans used a lot of bear fat (much more about this in a later post, check back). Bear fat can be relatively high in the polyunsaturated linoleic acid, similar to corn-fed lard. So if a native person is eating bear grease, SCD1 will be lowered to create an overall relatively saturated blend of body fat. The same thing is true of monounsaturated fats to a lesser degree.
But there are a number of unsaturated fats that inhibit SCD1 much more specifically and to a far greater degree.
Palmitoleic Acid
Thanks to Amber O’Hearn for turning me on to this one! She is working on a follow up post over at her blog mostly-fat.com.
Palmitoleic acid is an omega-7 monounsaturated fat that is the end product of the reaction of SCD1 with the saturated, 16 carbon palmitic acid. Like with so many enzymes in biochemical systems, SCD1 is inhibited by its own end-product. Generally this is referred to as end product inhibition. It is the cell’s way of saying, “Thank you, enzyme. You’ve done enough already.” A study in obese sheep showed that supplementation with 10mg/kg of body weight – the equivalent of 900 mg per day in a 200 lb human – prevented 77% of weight gain. Beef is an excellent source of palmitoleic acid, an 8 Oz ground beef patty has 1300 mg after cooking, according to the USDA.
If you want to use palmitoleic acid as a supplement, I recommend the more concentrated supplements purified from fish oil. Sea Buckthorn oil is often sold as a palmitoleic acid supplement. The berry has a lot of palmitoleic acid but is often mixed with the seed oil which is very high in linoleic acid. Additionally, Sea Buckthorn oil packs in a pharmaceutical equivalent amount of anti-oxidants, which will interfere with ROS signalling.
CLA
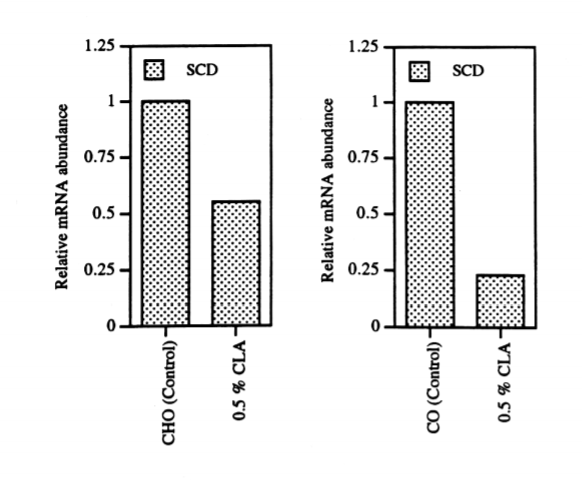
Conjugated Linoleic Acid (CLA) only differs from linoleic acid in the one of the two double bonds in the trans configuration. CLA is found in animal sources and is particularly high in grassfed dairy products. CLA supplemented at 0.5% of the total diet – the equivalent of 3 grams (3000 mg) per day in 200 lb humans – has been shown to reduce SCD1 expression by 45% in the liver of mice on a fat free diet and by 75% in the livers of mice with a 5% corn oil diet.4 It was also shown to elicit an impressive reduction in SCD1 levels in human liver cells.5 The CLA levels of full fat dairy products may go a long way towards explaining why full fat dairy products are associated with a reduction in obesity and diabetes rates.6
EPA and DHA
Fish oil is a combination of the long chain Omega-3 fats EPA and DHA, among others. It has shown an impressive reduction of SCD1 levels at a level of around 3 grams per day for a 200 lb human.7
Considering The Scenarios
A good theory of obesity should be able to explain all of the scenarios we see in the world. Now we’ve seen the science behind what SCD1 can do and the dietary factors that control its expression. Here is my summary of different scenarios of leaning and fattening diets and how SCD1 ties them altogether.
The high starch, very low fat diet from birth leads to leanness. A person living off of starch needs to produce some SCD1. They create body fat from starch by de novo lipogenesis, which only creates saturated fat. Saturated fat has a very high melting point and you cannot build cell membranes with proper fluidity, for instance, without a certain proportion of monounsaturated fats.
On the other hand, we know that a person living in China, eating a lifetime of starch has body fat that is quite saturated. The system regulates itself quite well. If a starch eating person packs on fat through feasting, etc, the increase in leptin drives an increase in fat oxidation which drives an increase in ROS which induces thermogenesis and the extra calories will be burned off.
The fat Native American living solely on maple sugar, living an ancestral lifestyle in 1770. Someone on a very low fat, high sugar diet has dramatically increased SCD1 expression – perhaps five to ten times higher than a starch eater. With that much SCD1, fat will be stored with a very low ratio of saturated fat to monounsaturated fat. When they gain weight and leptin tries to signal, they will fail to do leptin induced thermogenesis and become fat.
The lean hunter. A human hunter eating woolly mammoths or ruminants on the plains will not be doing De Novo Lipogenesis. Their body fat will come from their diet. It will be relatively saturated when ingested. The hunter will produce insulin when protein and fat are consumed together, which will stimulate SCD1. However, ruminant and mammoth fat also contain specific repressors of SCD1 activation such as palmitoleic acid, CLA, arachidonic acid and long chain Omega 3 and 6 fats, all of which are effective at suppressing SCD1. SCD1 levels will be very low, dietary fat will be stored as is. The person will be able to do leptin induced thermogenesis and remain lean.
The thin Parisian from 1970. A person living in a dairy culture that combines starch with butter is a blend of the starch eating person and the hunter. This person’s total response to SCD1 up-regulation from insulin plus carbohydrate will fall between those two. Their stored body fat will come mainly from dietary sources, the highly saturated butterfat. Pastured butter is a particularly potent source of CLA. The CLA will inhibit SCD1 up-regulation. The person’s bodyfat will remain saturated. They will be able to do leptin-induced thermogenesis and will remain lean.
That brings us to the modern fat American, combining starch with soybean oil. Paradoxically, the soybean oil and starch eating American has a lowered overall pressure to up-regulate SCD1 compared to the dairy eating person due to the SCD1 suppressing effect of polyunsaturated fat in the soybean oil. However, the specific fatty down-regulators of SCD1 – CLA, palmitoleic acid, arachidonic acid and others – are much more powerful down-regulators of SCD1 and soybean oil doesn’t have them. That’s problem number one.
Problem number two is that stored linoleic acid will start to cause a pathological inability to drive ROS production when cells switch from burning glucose to burning fat. ROS production in the mitochondria is the signal to the rest of the cell as to whether it is burning fat or glucose. A small amount of ROS tells the cell it’s burning glucose and SCD1 should be upregulated. A large amount of ROS tells the cell that fat is being burned it they should down-regulate SCD1.
When the stored body fat becomes high enough in polyunsaturated fat, in response to leptin the cell will increase fat oxidation which will drive a LITTLE ROS production. The cell will increase SCD1 in response. The metabolism is now broken. Leptin is doing the exact opposite of what it is supposed to do, leading to a positive feedback loop of unsaturation which leaves you with a post-obese physiology, even if you lose weight. This explains why very low fat, high starch diets haven’t proven effective for weight loss even though people on a very low fat, high starch diet their whole life remain lean. The post obese on a low fat diet are upregulating SCD1 when they burining dietary carbohydrate AND when they are burning stored fat. They can’t saturate their body fat from the starch because SCD1 is always present in high amounts.
They are stuck. They can’t do leptin induced thermogenesis. Their body temperature drops. They become fat. This is backed by the fact that when we look at the bodyfat of morbidly obese Americans, we see the trifecta: high PUFA, high MUFA and high SCD1.
High amounts of sugar probably aren’t helping Americans, either. Yet at the same time, the Parisian in the 70s also had very high sugar consumption. The French love jam and desert and Orangina. But the CLA in the croissants and fancy cheese apparently won the day.
Herbal Therapies
After you’ve fixed your diet by removing the vast here are a variety of herbs that can lower SCD1 activity that work by stimulating the MAPK and AMPK pathways. Yes, AMPK and MAPK different, I’m sorry.
Metformin
Metformin is a well known diabetes drug. It seems to have many modes of action, one of which is lowering SCD1.8 When SCD1 is lowered, liver cells store less fat.
Of course you need a prescription to get Metformin…
Berberine
Berberine is a Chinese herb Known for helping with fatty liver disease. Berberine is available without a prescription and inhibits SCD1 expression nearly as strongly as metformin! The paper is by the same team who did the Metformin experiment. 9 They repeated the same experiment in the same cell line with the same result. The only difference is that in this case they gave the cells fat to eat instead of glucose.
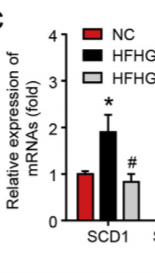
Berberine and CLA May Make a Potent Combo
G. pentaphyllum
G. pentaphyllum is another Chinese herb that, like berberine and metformin, seems to suppress SCD1 via the AMPK pathway.10 It has shown an impressive ability to prevent Western diet induced fat gain in the same mouse strain used in many of these studies. It has also been shown to reduce PPAR gamma levels, which is a direct up-regulator of SCD1 levels.
Hesperidin
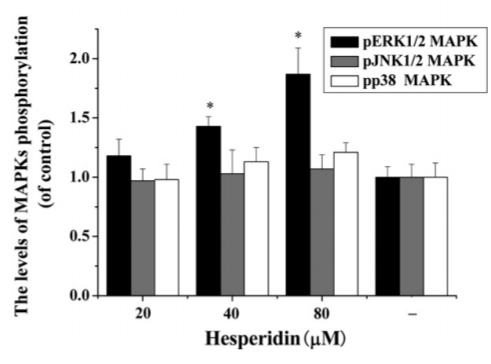
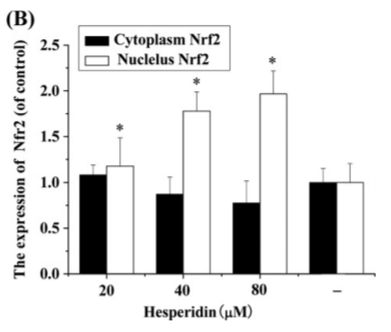
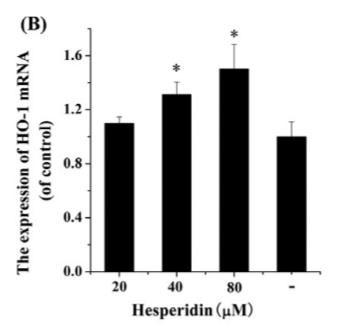
Hesperidin, a citrus extract, actually stimulates the exact same ERK1/2/MAPK signalling pathway that leptin does in leading to lowered SCD1 expression.11 Hesperidin also upregulates Nrf2 and HO-1.12,13 Nrf2 suppresses SCD1 expression and HO-1 increases uncoupling proteins which lead to thermogenesis.
A Natural Inhibitor
If you look at old pig feeding studies a funny thing starts to pop up. My 1951 copy of Morrison’s Feeds and Feeding reports, “When pigs are fed soybeans or peanuts, the fact that cottonseed meal tends to make hard fat may be of distinct advantage in counterracting the effect of feeds, such as soybeans and peanuts, which produce soft pork.” To which I always thought, “What?! Why would cottonseed meal produce hard fat? In 1951 the cottonseed meal still would’ve had a fat content of around 8% after oil extraction, most of which should be polyunsaturated.” Cottonseed meal SHOULD make soft pork, but it does the opposite. How is this possible?
It turns out that cottonseed oil contains a fat known as a cyclopropene fat. Sterculic acid, named after the tree Sterculia Foetida, also known as the Bastard Poon tree (I can’t make this up), is an 18 carbon length fat with a three carbon ring structure in the 9th position. Apparently, this ring structure – at the exact location where SCD1 is trying to desaturate – really sticks to SCD1 and gums up the works. Sterculia Foetida oil is around 70% sterculic acid. I recently had some imported and started taking a teaspoon full (4 grams) per day to see what would happen.
Of course I was concerned about safety. I wasn’t overly concerned because Sterculia nuts – which are around 50% oil by weight – are a traditional human food around the world. On the other hand, there is a study showing that mice lacking SCD1 who lack a functional LDL receptor have increased atherosclerosis and increased inflammatory markers, including an increase in IL-6. In addition, mice lacking SCD1 can develop a very bad skin condition presumably due to lack of sufficient oleic acid and have been shown to have increased ER stress.
I reasoned that mice lacking a functional cholesterol receptor is a very artificial model, but I also made sure to track my inflammatory markers. I also figured that since my body is CLEARLY full of oleic acid I won’t be getting into a deficit of it anytime soon! Also, a diet including butter, beef fat, low-PUFA pork and cocoa butter should provide a pretty fair amount of oleic acid. And I can always just stop taking it.
So I started taking it! My body temperature, which was typically around 97.0-97.2 in the days leading up to the experiment, normalized in about 5 days. I was so excited I took a picture of the thermometer at 98.6. I was doing leptin induced thermogenesis! I also have a metabolic testing device. Before I started taking the oil my resting energy expenditure (sitting on the couch) was around 2200 calories per day. A week later my heart rate – in the same position with the same heart rate – was over 2600 calories per day. I was doing leptin induced thermogenesis!! Another friend tried it and overnight she went from having a body temperature of around 97 to 98.7! A thrid friend saw a rise in body temperature after taking it for close to a month.
It’s too early to tell if this will lead to a weight loss miracle for me, although early results are promising. The scale is inching in the right direction and my waistline is decreasing. I expect this to work the way it did for Emmy. Small weight loss at first which will accelerate as my stored MUFA is slowly replaced by my mostly saturated dietary fat.
After two weeks on the oil, my inflammatory markers actually WERE up. Before the experiment, my C-Reactive Protein was 0.89, my TNF-alpha was 0.8 and my IL-6 was 0.7. Two weeks in they were at 3.04, 1.5 and 2.5, respectively. Am I worried about this? Not really. None of those numbers are cause for alarm, they’re fairly normal. My markers basically went from very low to high-average. If this helps me to fix my metabolism, I think it’s well worth dealing with short term mildly elevated inflammatory markers. Might I be worried about taking the SCD1 inhibitor if my inflammatory markers were very high to start? I might be.
You can always get your inflammation numbers tested with help from ownyourlabs.com, thanks to the great work of Dave Feldman and Siobhan Huggins.
In most studies, SCD1 inhibition has been shown to lower blood glucose but not in all. I haven’t seen a big change in mine.
Based on all of this I’m very excited to announce that I am having a small amount more oil imported and bottled and will be offering sterculia oil as a supplement. This is the first time SO will be available in the US, to my knowledge. A four ounce bottle should last 28 days at the recommended dose of 4 grams per day and will cost $39.99. There is a good chance that it will work at amounts as low as only 1 gram per day, but I’ve never tried it. It should begin to ship in January.
There have been several rodent studies on sterculia oil (SO). In mice lacking leptin, SO improved glucose tolerance but no effect on fat mass.14 The mice in that study were fed AIN-93G, a rodent diet consisting of purified corn starch, corn sugar, table sugar, casein and soybean oil, a perfect diet for fattening something up. Linoleic acid is about 8-9% of calories, the Goldilocks amount for fattening.
In obesity prone rats fed the same diet, sterculic oil improved glucose tolerance, decreased fasting blood glucose and reduced intra-abdominal fat mass.15 Still, the rats fed PUFA in addition to the SCD1 inhibitor were fat, showing that dietary linoleic acid can overwhelm the effects of inhibiting SCD1.
My favorite rodent study just came out in March of this year. The fed regular lab rats a standard, very low-fat, low-PUFA “chow” diet or the same diet with 30% pure fructose in their drinking water. Rats apparently love fructose water and massively increase caloric intake on it and become obese. The same rats, supplemented with 0.4% sterculia oil (SO) – the equivalent of 3 grams/day for a 200 lb adult – still have massively increased caloric intake and gain more fat compared to rats given regular water, but visceral fat gain is hugely reduced compared to rats given fructose with no SO.
| Low Fat Diet (LF) | LF + Fructose (LFF) | LFF + Sterculia Oil | |
| Calories | 402 | 757 | 705 |
| Fasting BG | 122 | 184 | 117 |
| Triglycerides | 97 | 203 | 65 |
| Insulin | 2.4 | 3.5 | 2.7 |
| Visceral Adipose Tissue | 3.2 | 5.4 | 3.9 |
Conclusion
Insulin and leptin are in a long term battle over the saturation level of your body fat. They do this by regulating the gene expression of Stearoyl-CoA desaturase, which turns saturated fats into monounsaturated fats. Insulin directly upregulates SCD1. Leptin acts by increasing fat oxidation in the mitochondria which drives ROS production. Very high levels of ROS increase Nrf2 activity and stimulate the ERK1/2 MAPK pathway, both of which down-regulate SCD1. This leads to your fat becoming more saturated which increases ROS production, setting in motion a positive feedback loop resulting in increased levels of thermogenic uncoupling proteins which allow you to burn off stored fat as heat.
If stored body fat becomes too unsaturated, either by ingestion of polyunsaturated fats (PUFA) or by constitutive up-regulation of SCD1 from – for instance – consuming an all maple sugar diet, this sets up a positive feedback loop in the opposite direction. Unsaturated fat produces less ROS and so the response to a leptin induced increase in oxidation is to increase SCD1. This leads to ever decreasing saturated fat levels and constantly up-regulated SCD1 resulting in a post obese physiology.
Strategies to fix this problem include eliminating all dietary unsaturated fats to the extent possible by replacing all sources of fat with dairy, beef suet, cocoa butter or stearic acid enhanced butteroil. This includes avoiding relatively unsaturated sources of fat such as nuts, avocados, olive oil, chicken fat and even most pork fat unless it is Low-PUFA pork. Certain polyunsaturated fats have the effect of dramatically reducing SCD, as do several herbal supplements. Sterculia oil is a natural inhibitor of SCD1 that seems to have a good safety profile and is a traditionally consumed human food.
- 1.Matsui H, Yokoyama T, Sekiguchi K, et al. Stearoyl-CoA Desaturase-1 (SCD1) Augments Saturated Fatty Acid-Induced Lipid Accumulation and Inhibits Apoptosis in Cardiac Myocytes. Stadler K, ed. PLoS ONE. Published online March 8, 2012:e33283. doi:10.1371/journal.pone.0033283
- 2.Dobromylskyj P. Protons (57): When Glucose Becomes Palmitate. Hyperlipid. Published August 2020. https://high-fat-nutrition.blogspot.com/2020/08/protons-57-when-glucose-becomes.html
- 3.Rojas IY, Moyer BJ, Ringelberg CS, Tomlinson CR. Reversal of obesity and liver steatosis in mice via inhibition of aryl hydrocarbon receptor and altered gene expression of CYP1B1, PPARα, SCD1, and osteopontin. Int J Obes. Published online January 7, 2020:948-963. doi:10.1038/s41366-019-0512-z
- 4.Lee KN, Pariza MW, Ntambi JM. Conjugated Linoleic Acid Decreases Hepatic Stearoyl-CoA Desaturase mRNA Expression. Biochemical and Biophysical Research Communications. Published online July 1998:817-821. doi:10.1006/bbrc.1998.8994
- 5.Choi Y, Park Y, Pariza MW, Ntambi JM. Regulation of Stearoyl-CoA Desaturase Activity by the trans-10,cis-12 Isomer of Conjugated Linoleic Acid in HepG2 Cells. Biochemical and Biophysical Research Communications. Published online June 2001:689-693. doi:10.1006/bbrc.2001.5036
- 6.Kratz M, Baars T, Guyenet S. The relationship between high-fat dairy consumption and obesity, cardiovascular, and metabolic disease. Eur J Nutr. Published online July 19, 2012:1-24. doi:10.1007/s00394-012-0418-1
- 7.Rossmeisl M, Medrikova D, van Schothorst EM, et al. Omega-3 phospholipids from fish suppress hepatic steatosis by integrated inhibition of biosynthetic pathways in dietary obese mice. Biochimica et Biophysica Acta (BBA) – Molecular and Cell Biology of Lipids. Published online February 2014:267-278. doi:10.1016/j.bbalip.2013.11.010
- 8.Zhu X, Yan H, Xia M, et al. Metformin attenuates triglyceride accumulation in HepG2 cells through decreasing stearyl-coenzyme A desaturase 1 expression. Lipids Health Dis. Published online May 14, 2018. doi:10.1186/s12944-018-0762-0
- 9.Zhu X, Bian H, Wang L, et al. Berberine attenuates nonalcoholic hepatic steatosis through the AMPK-SREBP-1c-SCD1 pathway. Free Radical Biology and Medicine. Published online September 2019:192-204. doi:10.1016/j.freeradbiomed.2019.06.019
- 10.Lee HS, Lim S-M, Jung JI, et al. Gynostemma Pentaphyllum Extract Ameliorates High-Fat Diet-Induced Obesity in C57BL/6N Mice by Upregulating SIRT1. Nutrients. Published online October 15, 2019:2475. doi:10.3390/nu11102475
- 11.Yumnam S, Park HS, Kim MK, et al. Hesperidin Induces Paraptosis Like Cell Death in Hepatoblatoma, HepG2 Cells: Involvement of ERK1/2 MAPK. Calvisi D, ed. PLoS ONE. Published online June 30, 2014:e101321. doi:10.1371/journal.pone.0101321
- 12.Heo S, Kim J, Choi Y, Ekanayake P, Ahn M, Shin T. Hesperidin improves motor disability in rat spinal cord injury through anti-inflammatory and antioxidant mechanism via Nrf-2/HO-1 pathway. Neurosci Lett. 2020;715:134619. doi:10.1016/j.neulet.2019.134619
- 13.Mahmoud AM, Mohammed HM, Khadrawy SM, Galaly SR. Hesperidin protects against chemically induced hepatocarcinogenesis via modulation of Nrf2/ARE/HO-1, PPARγ and TGF-β1/Smad3 signaling, and amelioration of oxidative stress and inflammation. Chemico-Biological Interactions. Published online November 2017:146-158. doi:10.1016/j.cbi.2017.09.015
- 14.Ortinau LC, Pickering RT, Nickelson KJ, et al. Sterculic Oil, a Natural SCD1 Inhibitor, Improves Glucose Tolerance in Obese ob/ob Mice. ISRN Endocrinology. Published online 2012:1-11. doi:10.5402/2012/947323
- 15.Ortinau LC, Nickelson KJ, Stromsdorfer KL, et al. Sterculic Oil, a natural inhibitor of SCD1, improves the metabolic state of obese OLETF rats. Obesity. Published online February 2013:344-352. doi:10.1002/oby.20040

I just read parts 1 and 2. Great work! Thanks so much for all that you’ve done on this site.
If I can only afford to take one of palmitoleic acid or CLA, do you have a guess about which one is more effective?
You said “Sugar up-regulates SCD1 much more dramatically than starch.” Do you think this is the main reason why sugar seems to be worse for health/obesity than starch? Are there other reasons?
Where you wrote, “Buried on page 11 of this great paper”, did you mean to link a paper?
Thank you!
Out of two I’d go with CLA, but I’m really just guessing. If I only took one of the supplementals, it might be bererine. But again, that’s kind of a guess as to what will work best and it likely varies person to person.
I think this is likely a main reason sugar behaves differently than starch. There are others – fructose goes straight to the liver whereas starch becomes blood glucose and can be taken up anywhere. (I think that’s also technically true of fructose as well but the majority goes to the liver.)
If you mouse over the superscript 1 at the end of the sentence, the link to the paper will pop up.
Brad
This blog is awesome. I just added berberine along with my daily heavy doses of Kerrygold butter and Cacao butter in my hot drinks, and on my low PUFA foods. My waist is shrinking rapidly. It actually makes me angry with the keto/carnivore community that demonizes starches. I’m much happier with my thin crust pizza with cheese and pepperoni, along with my ham and cheese croissants from the local French bakery. Keto killed my metabolism, and carnivore is boring AF. Thanks Brad!
You’re welcome! And thank you, I’m glad it’s working!
Brad
It seems like it is so easy to get CLA from the diet with TCD, so I would suggest supplementing with the palmitoleic acid, unless there is a reason that you don’t do high dairy intake.
Yes, this is a good point, – if you’re eating a lot of dairy it makes sense to supplement with palmitoleic acid. Conversely, if your eating a lot of fatty beef, it might make more sense to supplement with CLA.
Brad
This Fall I have especially concentrated on eating meat, very little carbs, and saturated animal fats. I do strength training concentrating on submaximal weights but significant load performance. I have noticed a loss of abdominal fat on the sides. In my daily supplements, I take berberine, CLA, and DHA/EPA. How much should I take? I weigh 90kg. Thanks!
I would suggest following the manufacturers suggestion as a starting point. It’s probably a good idea to test desaturase index before and after to see if you are getting the effects you want from the supplements.
Brad
Fascinating. Thank you for following up on this.
I have lost over 200# (am still morbidly obese), and am in that unenviable situation where even 1MAD, ketogenic, high-sat low-mono/pufa, moderate exercise, doesn’t seem to be doing much for reducing my bodyweight further. I’m absolutely open to pretty much anything that won’t get me arrested to continue reducing fat at least a little. Pre-ordered the oil to give it a try. I have Berberine. (Can’t afford anything else right now thanks to gov’t killing jobs/my industry in C19 response.) Will save the Berb for when I get the oil and try them at the same time.
Really appreciate your making this stuff so approachable for the layman. After your early posts I started melting cocoa butter, adding some cocoa, then putting that in silicone molds for little pieces I keep in a ziploc in the fridge and drop in my coffee to melt. Super satiating. I dropped eating pork entirely, chicken except breasts with fat removed, and added beef tallow and cocoa butter (mostly use coconut oil for cooking), ditching olive and avocado oil.
I wonder why you do not usually mention beef tallow as a sat fat? I got some and use it, it seems surprisingly not as hard as I expected. (I do see online that grass-fed is higher in stearic than ordinary beef tallow.) Do you have thoughts on tallow?
Thank you!
I would think that taking berberine and sterculic oil together might be redundant. If it were me, I would start taking the berberine now, then switch over to sterculia oil when you get it. Save the rest of the berberine for when you’ve lost the weight and want to come off of the oil.
Yes, tallow isn’t that saturated compared to dairy or suet or cocoa butter. I’m not saying it’s a bad fat, it’s just that if you’re trying to use it to firm up the blend going into your mitochondrial to drive ROS generation, you might want something stiffer.
Brad
Brad,
I recently rendered my own tallow from some suet I purchased from a shop that sells local beef. Is that going to be higher in sat. fat than tallow rendered from the body fat? I was under the impression this was pretty saturated. I sure hope so. I’ve been adding a tbs to my ground beef when I brown it (I don’t drain off the extra) and the other day I fried some beef liver nuggets. I used whey protein powder and seasoning as the breading. Came out fantastic.
I also mixed in a bit of your stearic acid powder as I was jarring my tallow to up the payload. My own variation of your butter oil. But if it’s not very high in sat. fat I’ll use it up then not make any more.
Thanks,
Casey B
The tallow from suet should be MUCH more saturated than other beef fats!
Brad
Hey Brad-
Thank you so much for all this amazing and informative content.
Sorry to ask a question to which the answer should be obvious. Do I take from your article that fish oil is safe to take along with this whole system? I’m taking Nordic Naturals Ultimate Omega but just one capsule. Just asking because I seem to remember seeing somewhere (maybe on Reddit) that it was discouraged.
I’m a 75 yo woman with a life long weight problem – right now only over by approximately 20 pounds. I’ve been over by as much as ~100 pounds and have only been able to lose with the help of The hCG Diet (lost 92 pounds in 2009-2010). It’s the only thing that tempers my appetite. I’ve used it several times since to keep at least most of the weight off. I do tend to overeat. I’ve tried keto and carnivore and mostly stay low carb when not doing one of those. The TCD resulted in gain in my case, probably due to eating too much homemade sourdough bread. With loads of Kerrygold, of course. I ordered some sterculia oil and am looking forward to giving that a try. By the way, I’ve been speculating for years as to how hCG works and have figured that it has something to do with leptin, but now I’m wondering if it might have an effect on scd-1. A quick Google search didn’t come up with anything obvious.
Thanks again.
Hi Dana!
Forgive my ignorance. What is the hCG diet?
Brad
Hi Brad –
hCG (human chorionic gonadotropin) is a hormone present in pregnancy (it’s the substance tested for in pregnancy tests). In the 1950s, A. T. Simeons, MD, of Rome, Italy, developed a weight loss diet using low doses of hCG. He wrote a book about it, pounds & Inches, copyright 1971, that is still available on Amazon Kindle (someone republished it from his original work). In his day, he treated people from all over the world. I remember hearing about it in the 1970s when clinics sprung up in the US. The diet consists of a strict 500 calorie diet of very specific foods. hCG generally suppresses one’s appetite. It also, according to Simeons, has an effect on the hypothalamus. A you might imagine, it’s not necessarily the healthiest way to lose weight, but it works for me. I do modify it a bit from the original protocol. BTW, it is used by both women and men.
If I could venture a guess here, Dana (also having done multiple rounds of hCG)….I felt fantastic on the diet and it was also the only thing that allowed me to drop weight. I thought the diet was extreme but I felt absolutely miserable and had been to many doctors so thought “Why the hell not??
This is no way to live.” Within 3 days of hCG on multiple rounds, my energy would ZOOM upwards and I would start to feel fantastic. Then I learned, when I dropped the hCG and followed the same diet, I found I had nearly similar results. I started my weight loss journey 8 years ago and now hover at a much lower weight than ever (about 30 pounds away from my ideal weight, but 40 pounds down from pre-hCG). Today, I eat OMAD Carnivore (all beef mostly) but I really have to limit the AMOUNT of food I eat to keep the weight dropping (which is not really sustainable). The minute I switch off of Carnivore, the weight comes back easily (20 pounds in a month or almost 1 pound a day). On this WOE, I monitor my progress by checking my blood sugar (morning fasting and right before my one meal). I feel that the key to my weight loss is getting blood sugar as low as possible. This seems to be in line with this blog post as getting insulin down seem to down-regulate SCD1 (not sure the terminology is right there).
Would love to hear more of your thoughts on this Dana.
Interesting. I wonder how much is known about its affects on the metabolism.
Brad
So is the goal of the up/down-regulation of SCD1 to maintain a particular cell membrane fluidity? Is there research on the particular tipping point/storage point in terms of excess PUFAs i.e. eating PUFA that lowers SCD1, burning stored PUFA that raises SCD1? Conversely, can eating excess saturated fat up-regulate SCD1 or is the effect of oxidating stored saturated fat a net down-regulation of SCD1? Are the specialized polyunsaturated fats i.e. CLA stored? I’m just trying to make sure I have the concept straight, appreciate all your posts!
Yes! Thanks for this. The regulation of SCD1 is paradoxical in a lot of ways.
You say the “goal” of SCD1. Let’s put that in context. The organism NEEDS SCD1 for membrane fluidity if its living on pure starch.
If the organism only makes as much SCD1 as necessary for that process in the case of a starch diet. When sugar or unsaturated fat (most lard, chicken fat, nuts, olive oil, avocados, vegetable oil) are consumed is where things get dicey!
Maybe the “dieticians” are half right. Eat complex carbs BUT also SFA?
https://www.google.com/url?sa=t&source=web&rct=j&url=https://www.mdpi.com/2073-4409/9/1/140/pdf&ved=2ahUKEwjPyKXs9IrtAhVBCc0KHboGCRoQFjAAegQIARAB&usg=AOvVaw2qqHnNv45uJfOwLvmZEISI
Great stuff as usual! Another good reference above. Have you looked into methylene blue?
Rich
Article link is very comprehensive. Brad should look into it. downsides of effective SCD1 inhibition. So people need to be care and vigilant.
Methylene blue An electron donor to ETC, supposedly facilitating mito function
OxLDL inhibits SCD1 according to above article
Fascinating blogs. I’ve learned so much from you Brad.
I’ve been carnivore for the last 3 years with great results. However I’ve gained substantial weight over the last 4 months. The only difference was that I got hooked on eating Wagyu beef ribeyes. Because of your blogs, I did some research and recently learnt that wagyu beef has much more oleic acid fat because the cows have been genetically modified to convert saturated fat into monounsaturated fat. And of course this is marketed as “more healthy” mufa meat. It is very expensive and tasty, but it is now my #1 suspect of why I’ve fattened up. My waist size went from 35 to 40 in just 4 months. Brad – would you agree that wagyu beef might be the culprit ?
OF COURSE!! Think about it. The only way to get the fat into the muscle fibers of the wagyu beef is that the Wagyu muscles fibers are overexpressing SCD1 and therefore the fat in that beef will be highly unsaturated!
That’s probably not the fat you want to be eating!
That may explain why Carl, of 2KetoDudes, a few years back, had stubborn excess weight despite regular 7-day fasts, coupled with hours-long cycle rides. He often remarked on the Wagyu steaks from Costco. It’s one of the reasons I didn’t by into the long-day fasts.
It wasn’t until my 40’s I gave into the hype of low-fat milk, which is when my weight crept up and health issues developed. Despite growing up in Corning, which is surrounded by dairy farms, whole milk and butter were a mainstay. My Dad (Wellsboro PA) remarks that growing up, any sandwich both slices of bread were heavily buttered. He and his three older brothers were tall and lean, the two youngest, kids during WWII, were short and fat, as they ate the margarine sandwiches during the war.
WHAT? You’re in Corning?! Right down the road 🙂
My family is from dairy farmers in Wayne County, NY. I have family photos from ~1980 showing all very lean kids/moms/dads/etc. Today: not so much!
They definitely were eating a lot of full fat dairy before 1980.
Brad
Thank you so much – it all makes sense now from the ROS and SCD1 mechanisms that you have explained so clearly.
I am disappointed at myself for falling for the marketing hype on these types of expensive meats. But I’m so glad I found your blog and I’ve been binge listening to all your recent podcast appearances. Better later than never!
Over the last two weeks I’ve been enjoying bulletproof coffee with your stearic acid butteroil. I will eliminate the wagyu beef, and go for lean grass-fed beef cuts with added butter/butteroil instead. Will keep you posted, I hope I can reverse this self-inflicted problem!
Damn it! I just got some beef tallow to fill my deep fryer, and they were out of grass finished beef tallow, so they “upgraded” me to waygu beef tallow! Now I shall have to send it all back (haven’t opened one jar yet, thankfully).
I’m so glad I read this blog before filling my fryer up with possibly MUFA tallow!
Thanks!
Hi, I tried to pre-order the Sterculia oil, but couldn’t complete the order. It said no shipping info to my area (I live in Canada.). Is there anyway I can order. Thanks, Margaret
Hi! Doh! I am having major problems with the shipping software… Check back in a week or so.
I switched from olive oil to macadamia oil for salads and some for cooking or adding to starch. Is that a bad move?
Do sprouted seeds like pumpkin or sunlower have better profiles than non sprouted.
Could you make some cheat sheet template of how to analyze omega quant results?
Look for my upcoming article on how DIETARY monounsaturated fat is different from mono generated in the fat cells.
To determine Desaturase Index, just divide Oleic acid (18:1n9) by stearic (18:0).
Brad
Brad, I am truly amazed by your research, you are inspiring me so much! (From an humble biotech student).
I found this study on PubMed: http://qr.w69b.com/g/r0nJ82blK which basically says that overfeeding muffins made with saturated fats created all the effects that we see in high PUFA diets. They claim that sunflower oil rich muffins made the participants gain muscle. I see the little dirty paws of the american diabetes association, but I’m still surprised by the results. The saturated fat used was palm oil (which still has PUFAS), but I can’t grasp how those results were possible
Hi Edoardo,
Ha! You’ve scooped me. This series of articles is leading up to that study. Explaining that study will be a sort of grand finale. I haven’t figured it all out yet, but I’ll give you a sneak preview. In people eating sunflower oil muffins the weight of their muscle tissue increased because they were being purposefully overfed with muffins. If you had a guess would you guess their muscle mass increased because:
1) They were adding muscle protein.
2) They were adding muscle fat.
Brad
Brad, do you think the slight increase in your inflammatory markers is like having induced a fever to rev up your metabolism? By the way, just received 3 bags of stearic acid via my shipping service down here in Ecuador where I’m a retired US expat. I’m lucky Ecuador sells metformin without a Rx. I put myself on 500mg at night for longevity and now thanks to your research I have another reason to stick with it. Also I can walk into a lab and order tests without a doctor. Sometimes it’s great not to live in the US!
I’m honestly not sure the inflammatory markers have anything to do with the sterculia oil. It could just as well be due to my recent insomnia or any number of other factors.
Brad
That’s good because if it is the oil, I don’t think taking the oil in the middle of a pandemic where high inflammation seems to be bad is a good thing.
The highest HS-CRP I got was 1.7, after fasting 4.5 days. But it’s also something for which I did not initially test.
Hello! What is the difference between cocoa butter and cacao butter? Is using the wrong line detrimental?
My understanding is that they are the same.
Brad
We need 1000 people to take this oleic/stearic test, and hydrostatic weighing to see how tight the correlation is.
Agreed! I’m talking to Dave Feldman about doing a project with the Citizen Science Foundation.
Brad
Amazing post Brad.. one of the best!!
Regarding the DHA supplement, Peter lately is very sceptical with them, even writing that fish oil massively fattens the liver in rats.
Don’t you see a problem with them?
And regarding berberine, what would you do if you were an athlete trying to build muscle and athletic performance… Would you take it or would you be worried that it blunts the anabolism?
Regarding the sterculia, what’s the mechanism of harm? Why does it increase inflammatory markers?
Thanks again for your amazing work!!
I think that DHA in large amounts is probably bad. A couple hundred milligrams can help knock down SCD1, I wouldn’t do more.
I don’t THINK berberine would affect anabolism by stimulating AMPK, but I also don’t know enough about it to say.
As to sterculic, my guess is that the mechanism of harm might be oleic acid deficiency. Clearly I don’t have that. It’s totally likely that my inflammatory markers are up (a little) purely due to insomnia or other random reasons. It’s also possible that something happened to REDUCE my markers the day I took my baseline numbers. Either way, I don’t see anything yet to be alarmed about.
Brad
hey brad thanks for your post
i have a question. i currently feel a small lack satiety after my mostly carnivore meals, presumably because my body is trying to gain some weight back (i have some empty adipocytes i guess)
would berberine help with this particular problem?
thanks
I can’t really say if Berberine would help with satiety. For me having a potato with my steak helps with satiety 🙂
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5537804/
Hey Brad
LOVE your work .. I work with menopausal women where weight gain is a truley bothersome symptom..
the above link ia about valerian which seems also to down regulate SCD1..
all best and I will donate!! Sarah
Interesting study, unfortunately you cannot determine the amount of valerian was given. They list how they made the extract but forgot to mention how much it would yield. So there is no telling how much valerian is needed in order to get the same results. 1% an unknown extract per food given by weight, or 10 grams of extract per 1KG of food (high fat diet w 60% fat, by weight, not calories). For a carnivore diet that is 195 g of meat and 300 g of fat per day, plus 5 g of valerian extract. That is a lot of extract for one person. Or 195 grams of fat free bread with 300g of fat, plus 5g of Valerian. That much fat will put you off food by itself and probably cause weight loss, if the right fat is used. Such a high dose of valerian extract… not sure that is advisable.
Traditionally Valerian root extract is considered to be an appetite stimulant. An excessive dose can cause headaches, nausea and vomiting. Perhaps the reason for the decreased food intake? It is also something that should not be taken long term as it can cause you to experience the exact opposite of the sought after therapeutic results – being excitable, uneasy and unable to sleep. So overall a study, although with obesity in people mind, that is not really useful in a practical application.
If your sleep quality is poor valerian extract would be much better served helping you get a good night’s sleep. Improved sleep quality can help with weight loss. Or if you are anxious and eat constantly because of that. Either way, only a short term solution.
Hi
Saw this reference from the link about people traditionally eating the wild almonds (Kalumpang http://www.stuartxchange.org/Kalumpang.html)
Delay of Sexual Maturity / Oil: Study evaluated 21-day old female albino rats of Sprague-Dawley strains on diets partially substituted with S. foetida oil. Results showed 3% oil in the diet, which supplied about 105 mg of sterculic acid per day definitely delayed the sexual maturity of female rat by criteria of age at the time of vaginal opening and regularity and length of consecutive cycles. (22)
And wonder about the safely for women in taking Sterculic Acid?
I definitely wouldn’t recommend taking anything near 3% of diet as S. Foetida oil. 0.5-1% is enough. Nor do I recommend giving it to pre-pubescent children. As to adult women. I’d guess it’s safe but I wouldn’t take it if I was trying to have a child, nor would I take a lot of other supplements if I was trying to have a child. I just think there are too many unknows.
Having said that, check back in over the next couple of weeks, I have an upcoming post on infant obesity to which this comment has some relevance.
Brad
Hello Brad, Such a great work. This work nailed it. I am in a 1.5 year strict carnivore journey while lost 30 pounds but find myself in a weight loss challenge since last 6 months. This work explains why. Just a bit of honey or fruit, will massively increase wight gain and water retention. I avoid them. However I am still having my morning coffee.
What is your take of the effect of black coffee, caffein on SCD1?
I am wearing CGM and what I see is: When I have it in a morning, 4 espresso, my blood sugar rise from base line of around 100mg/dl to about 140mg/dl ( gluconeogenesis liver) and sugar staying up for about 8 hours, which explain temporary insulin resistance ( dont know which one: muscle or fat tissue or both) due to caffeine.
My base line insulin is around 10 fasting.
Many thanks
I haven’t even considered yet the effect of coffee (or alcohol) yet on SCD1 levels. which is funny because I am a HUGE coffee (and wine) consumer.
I think it’s been less studied but it’s definitely on my radar screen. In the meantime, I would say that I wouldn’t expect a short term change in SCD1 levels to have an effect on blood sugar levels. So I doubt the short term effects from coffee you’re seeing on blood sugar levels would be due to SCD1 changes. I’m open to being wrong.
Brad
You’re likely experiencing a stress response when drinking black coffee while fasted.
Since you have a cgm you can run an easy experiment. Try coffee with cream, then try coffee with cream and sugar. My bet is you will find your highest blood glucose will be with black coffee.
Brad,
I just read that American’s body temperature has dropped from 98.6F to 97.5F. See link below. Do you think this could be accounted for by metabolic down-regulation by high-PUFA diets?
https://www.news.ucsb.edu/2020/020081/drop-temperature
YES!
I don’t just THINK this, I BELIEVE this. I have an upcoming post about it…
Brad
Hi, Brad. I’m not sure if you’ve addressed this more thoroughly somewhere and I haven’t seen it (if so, my apologies!), but I have a question: I first ordered your butteroil and tried TCD at the beginning of the year. Sadly, being a menopausal woman, I quickly began to gain weight instead of losing it. I stopped as soon as I saw it was only going to continue. At that time, I was including starches with the SFA. Now that I’m seeing this post, I’m wondering if trying to do TCD Carnivore style like Emmy might work for me instead? And if I add in Berberine (as I’m sure much of my stored fat is unsaturated!)? Do you have any other advice for those of us who gained on TCD? I’m struggling to lose even a few pounds, and I have plenty to lose, so it’s very frustrating. Thanks so much, your blog is a breath of fresh air compared to everything else out there!
Hi Lisa!
Clearly I can’t say for sure what will work for you… I am getting some – VERY EARLY, TOO SOON TO TELL – good reports from people trying CLA. I think a combo of CLA and berberine makes a lot of sense to try to knock down SCD1 levels if you gained with TCD. Also, check the blog next week, I have one more idea but I’m still reading through the literature….
Brad
Thank you! I’m trying the berberine for now; forgot to mention that with the Keto/Carnivore, I am including plenty of red meat and dairy. The beef is not grass fed, but the butter (and sometimes cheese) is. So I was thinking I have the CLA part covered in that aspect, correct? Also started adding Cacao Butter wafers to my coffee again. If Berberine doesn’t seem to help after a month or so, I may try the AMPK supplement. Looking forward to that next blog post, thanks again!
This is fascinating – thanks very much for putting this together! I’m curious about the state of being “post obese”. Is it the case that this state is completely defined by the ratio of stearic acid to oleic acid in tissues? If so, which tissues? Most importantly, what are the most effective ways to shift that ratio, and how long would it take to make meaningful progress?
Thanks again.
I’d say it’s DEFINED BY up regulated SCD1. The ratio of oleic to stearic acid is simply an indirect way of measuring your personal level of SCD1.
I’ve presented what I know about controlling SCD1, but check out the article about dietary monounsaturated fats coming next week.
Brad
Great stuff Brad — you are really putting the puzzle pieces together! Keep up the good work!
Thank you!
Brad,
Thanks for another great article!
Two quick questions:
1) If someone is already on Metformin, would Berberine be redundant or would it have independent benefits?
2) Is there a role for MCT oil in replacing PUFA”s/MUFA’s?
Thanks
Brian
Hi Brian!
1) My guess is they’d be redundant.
2) I think they can play a role but I have as many questions about them as I have answers… I prefer palmitic and stearic acid, but you can’t make salad dressing with them!
Brad
Im confused. You said “It turns out that soybean oil contains a fat known as a cyclopropene fat.” Is this correct or did you mean to say “It turns out that cottonseed oil contains a fat known as a cyclopropene fat.”?
Thanks
Yeah, I meant cottonseed oil… I’ve been meaning to fix it.
my oral temps are low for me now….97 or less. Desaturate index 2.0. No seed oils, but Macadamia nut oil occasionally swapped from Olive oil. Are you aware of any references regarding animal models showing activation of BAT (like PET FDG) by any of these supplements, especially sterculin oil?
Unfortunately there have only been three papers looking at sterculia oil to my knowledge and none of them have looked at the effects on uncoupling proteins or any other distinctive markers of BAT.
There have been other studies using OTHER methods of eliminating SCD1 that DO show distint upregulation of uncoupling proteins. The following link used antisense RNA to knock down SCD1 expression which led to increased levels of UCP1, 2 and 3 in BAT after 15 weeks.
https://www.jci.org/articles/view/23962/pdf
Berberine HAS been to shown to upregulate UCPs. For instance:
https://bpspubs.onlinelibrary.wiley.com/doi/full/10.1111/bph.12666
Brad
I am trained to avoid safflower oil so seeing that CLA supplements are mainly from safflower oil is worrying.
Because you are supplementing sterculic acid, is it the case that grass fed butter and ghee and high-fat cheeses like feta are not rich enough in CLA to downregulate SCD1 enough in the typical American post-obese? Would it be reasonable for me to try adding high-fat, grass fed dairy to a grass-fed beef (lots of suet, but not dairy) heavy carnivore diet, specifically for the CLA content? If so, is targeting a body temperature of 98.7 F the best way to verify that this is working?
Yes, the CLA supplements are from safflower oil, which of course I don’t recommend in general, but in this case they’ve purified out just (ok, mostly) the CLA, of which there are two common isomers and this is a 50:50 blend of them. So fr I’ve heard good reports from people trying them.
I think adding full fat grass-fed dairy to a carnivore diet could indeed be helpful. Then you’d have palmitoleic acid from the beef and CLA from the dairy which would be a nice one-two punch. Will that be enough to fix any individual? Hard to say ahead of time.
Best ways to know what’s working are probably monitoring body temperature and testing your red blood cell Desaturase Index.
Brad
Hi Brad,
Do u have any opinion on what would cause exclusively visceral and or facial fat in someone? (I dont mean lipodystrophy kind of lack of sub Q fat – just pretty lean overall but huge visceral and facial adiposity
Hi Brad,
Do u have any opinion on what would cause exclusively visceral and or facial fat in someone? (I dont mean lipodystrophy kind of lack of sub Q fat – just pretty lean overall but huge visceral and facial adiposity.
Also, what dosages of berberine are we talking about? Or what’s the range you would recommend for a) someone who’s metabolism is little broken and b) someone who’s metabolism is terrible ruined?
THANK YOU MUCH FOR ALL THOSE GREAT POSTS!
It seems there’s a fairly strong genetic component to visceral fat. For instance, Indians (people from India, not native Americans) tend to have low BMI but visceral adiposity.
As to berberine, I would say to start with the manufactureres recommendation and go from there. I’m sorry I don’t have any more specific recommendation.
Thank you!
Brad
Thanks for this enlightening read.
Have you listened to Saladinos podcast with Peter? In that Peter explains, that obese people, when they release fatty acids, they are SATURATED fatty acids.
And when you go on keto diet, the fatty acids released are more unsaturated, even PUFAs.
How do you explain that, especially that obese release SFA?
I DID listen to that. I need to go back and listen again. I honestly don’t have an explanation for this at the moment, but it’s interesting.
If obese release SFA, would that then mean that what is left in the fat cells is less saturated? And if you release PUFA on keto diet, then what is left in the fat cells is more saturated? Would that actually fit with the finding that the fat of obese people is more unsaturated?
Well writen and incredible well researched and put together. Thank you Brad.
2 questions arose by reading the comments:
1. is eating fatty beef also kind of problematic if its not suet but rather ground beef? (i mean should one prefer leaner beef and increase butter instead?)
2. does a beef heavy diet (2lb groundbeef per day) in combination with lots of milk and butter (0,25lb per day) provide enough CLA and palmitoleic acid or could one benefit by extra supplementation?
Thank you!
1) This week I’m going to publish a post on the effects of MUFA, which will help put the ground beef argument into context.
2) Perhaps but it’s hard to know in any individual. A cheap way to test this is your resting body temperature. It should be over 98. Also, you can test your desaturase index. I’m hoping to publish more people’s data as references as they come in. I’m also a believer in hitting SCD1 through multiple pathways. The CLA and palmitoleic might be plenty but it still could make sense to add berberine or hesperidin.
Brad
That post is up, BTW!
Dietary Monounsaturated Fat, The SCD1 Theory of Obesity, Part 3
Brad
WOW! This the best blog on the internet!! Props to you.
Im not sure though on visceral fat.. whats you opinion on this?
Is visceral fat something special compared to sub Q? I mean, does it behave the same as sub Q fat? (especially regarding the information in your blog… )
Or a there certain triggers that preferentially increase visceral fat compared to sub Q fat?
Hey, thanks!
One of the primary ways that visceral and SubQ fat differ is that visceral fat has a higher expression of LPL and is therefore more directly targeted by dietary fat. That is why some people see an immediate drop in waistline when they consume a lot of long chain saturated fat.
Brad
Hi Brad!
Thanks for the SCD1 series, which nicely answers my earlier question about why the mice on stearic acid ate more but still lost weight.
I have been reading through the new series a few times, understanding a bit more at each pass. However, there is one point where I am stuck:
“When sufficiently saturated stored body fat is being burned at the request of leptin, lots of ROS is produced and SCD1 is inhibited, initiating the cycle of leptin induced thermogenesis. BUT! If that stored body fat has enough PUFA in it, ROS production will be insufficient to drive enough ROS production to shut down SCD1 and in fact SCD1 will be increased!
….
Burning PUFA in the mitochondria looks to the rest of the cell as if the mitochondria is burning glucose. The level of ROS production is a signal from the mitochondria to the rest of the cell to tell the nucleus in which phase of the Randle cycle its in – whether it’s burning fat or glucose – so that the nucleus can respond appropriately. The cell does the appropriate thing in the context of a small increase in ROS production: it increases SCD1. Except that in this case the stored fat is already highly unsaturated. More SCD1 is produced and the fat gets even more unsaturated.”
I get from the above that burning more saturated fat -> more ROS -> less SCD1 and,
more unsaturated -> less ROS -> more SCD1.
But then I am confused when I arrive at the “Unsaturates” section, which states that:
“All unsaturated fats lower SCD1 production, polyunsaturated fat more than monounsaturated fat.”
This seems to contradict what I understood earlier. I am obviously missing something. Could you help me to fit the pieces together?
Yes.. It’s a bit confusing. There are things that affect SCD1 regulation directly. This would include things like starch, sugar and saturated fat, which upregulate it and unsaturated fat, which downregulate it. This is the body trying to achieve balance.
When leptin rises (ie you’ve gotten fat), what’s supposed to happen is that if your stored fat is properly saturated, the leptin increases fat oxidation rates which leads to the surge of ROS which should trump all of the other factors.
Brad
Thanks for taking the time respond to all these questions your readers have! And sorry for remaining confused.
I try to pinpoint better what I do not seem to understand. Your reply would have made immediate sense to me if you would have had “saturated” and “unsaturated” in the other way around like this:
“This would include things like starch, sugar and UNsaturated fat, which upregulate it and SATURATED fat, which downregulate it.”
What I am wondering is that how the saturated fat can both upregulate and downregulate SCD1? Is there something context dependent that I do not realise, like burning dietary vs body saturated fat?
Yes! So the regulation has multiple layers. Things like starch and saturated fat act on the first layer and raise SCD1 levels a little bit. The higher levels of regulation – especially things like erk1/2 signalling and Nrf2 signalling, both of which are controlled by ROS production and therefore are stimulated by leptin – usually trump the first layer regulators.
So if you are lean then eating some highly saturated fat like beef suet will cause SCD1 to go up a bit because as humans we don’t want our body fat to be as saturated as beef suet. Remember, the body is trying to achieve balance.
But if you are fat and you eat beef suet, the leptin (in a metabollically healthy person who has gotten fat because it is feasting season, for instance) will drive increased fat burning, which generates ROS, which stimulates erk1/2 and Nrf2 and SCD1 levels will remain very low. This is actually very logical because if you’re fat it’s likely that your bodyfat is very unsaturated. Again, the body is trying to achieve balance.
Brad
Ha! Thanks a ton for this. I think I got this now!
Hello Brad. Are you hoping that after some period of sterculia and berberine, the metabolism will eventually be restored to the “pre-obese” state with the correct SCD1 modulation returning?
And although its an extreme solution, would severe fasting to low total body fat levels followed by gorging on saturated fat also do the trick? Seems that if you just burned off nearly all the body’s fat then ate a hypercaloric diet of saturated fat for a period, the mitochondria would be burning mostly dietary fat, keeping SCD1 low, while the adipose was restocking with mostly saturated fat. Not a pleasant method, but it seems consistent with your SCD1 hypothesis.
This thing told me I am posting too quickly but the issue is I put in the wrong email address and sent again.
The problem with this theory is that fasting increases SCD1. A better option might be to consume a high fat saturated fat diet (or even a pure starch/fructose/ethanol diet) with proper inhibitors and/or down-regulators in place to steadily dilute out the stored unsaturates. Time will tell the better option, I suppose.
Brad
Hi Brad,
Whats you opinion on CoQ10 supplements?
If they would truly increase CoQ levels in the mitochondria, wouldnt they be highly fattening because less ROS are produced?
Yeah, I wouldn’t recommend them for that reason. Perhaps they could help if you were a type 2 diabetic with true mitochondrial oxidative stress, but that’s a whole other discussion.
Brad
Hi Brad. Great series! Answers so many questions. What do you you think about long fasts to expedite pufa removal?
Whether a long term one (30 days) or multiple short terms (7 days), to burn up bodyfat and eliminate pufas quickly, then rebuild the right way with the correct food and saturated fats.
Ive noticed so many people who try this route and even Ray Peat stuff. A lot of them just gain weight and get frustrated.
Peter over at hyperlipid said it took him 6 months before his energy went through the roof and see most of the benefits. Im assuming it was because his body was slowly replacing his fat from pufa to saturated.
So maybe suffering short term and fasting yourself down to a lean weight and then rebuildIng with the right foods and fats so now you have a more efficient body. Like a whole body rebirth almost. 🙂 as if you are starting with a “clean” slate.
You could also do a protein sparing modified fast where its just protein for 30 days. Take your SCD1 inhibitors to counter the insulin. Seen people drop 30 pounds in 30 days in the bodybuilding space doing this (they just do protein shakes for 30 days with extra nutrients added in). Its the after that kills them. They gorge on all the wrong foods and fat and just end up worse off then before.
If you have ever watched the show Alone it shows how quick protein only diets strips off body fat. One guy killed a moose and had his fat stolen by a wolverine. And despite having 100s of pounds of lean meat he was dropping weight so fast he was at risk of losing the show for medical reasons.
Just a thought for those who struggle because their fatty acid ratios are so off. Suffer short term so you can then enjoy an amazing diet that you know you can stick to for once.
Thoughts?
Hi! Interesting suggestions. As to long term fasting to eliminate fat…
The Feasting Mimicking Diet
Perhaps, I’m not sure. I suspect many lack the willpower and I worry about lowered metabolic rate. Also, fasting increases SCD1, putting your body into fat storage mode.
I think once the holidays are past I’m going to try something along the lines of The Feasting Mimicking Diet again while taking sterculia oil, with the idea that maximum dietary saturated fat will maximize fat flux, therefore replacing stored fats with dietary fats.
Brad
I am wondering you opinion on Camel Hump Fat as it appears to contain CLA. I am allergic to dairy and adverse to taking a CLA pill. Is it possible that Camel Hump Fat could be substituted for butter or would the high oleic acid make it not beneficial?
Interesting suggestion. This is the first time I’ve considered camel hump fat. It DOES have a very good overall fat composition – about two thirds saturated and one third MUFA. That’s comparable to butter, beef suet or cocoa butter. The CLA content is pretty good at 0.5%, which is less than grassfed butter, but higher than many things. It seems like a good option.
Brad
Awesome post, Brad.
Insulin and leptin are “master control hormones” – while testosterone (that everyone focuses on) is a “downstream hormone”.
Insulin and leptin are running the show, especially for fat loss.
Hi Brad .. regarding bodytemperature to assess SCD1 activity: I wonder why obese people are generally way less cold than lean individuals often not needing more than a thin wind jacket in snowing winter conditions. Is it just the fat that acts as isolation independent of lower body temp?
Well, one thing to consider is that an obese person has a higher body mass to surface area ratio. So proportionally they have way less surface area exposed to the cold.
Hello everybody hello Brad.
Amazing information in that article. Has been an eye opener.
But I missed one point: you write that burning PUFA looks to the cell as if it’s burning glucose (because of equally little ROS) to which the appropriate reaction is increasing SCD1 … Why is that? Why is burning glucose a signal to increase SCD1?
Maybe this passage from Brad’s post is helpful
“ A person living on a high starch diet – white rice – has to produce some SCD1. They need body fat for building cell membranes, fatty tissues, etc. They have to make this from starch and a certain amount of MUFA is required in that case for proper membrane fluidity.”
This is because if a person lives on starch their body fat comes entirely from de novo lipogenesis, which can ONLY produce saturated fat. Pure saturated fat is not fluid enough to build membranes, etc out of. So you need to make some MUFA if you live on starch.
If one was already decently lean (flat stomach but no abs) and already low PUFA, but struggling with some other health issues, would trying to get a desaturase index close to 0.9 be helpful? Maybe a month on Sterculia Oil and stricter ratios? Or would I deplete my oleic acid?
Thanks.
Perhaps… First off, it’s not entirely clear to me that the proprietary OmegaQuant test is exactly comparable to the methods used in The China Health Study. So it’s a little unclear whether 0.9 is a reasonable target goal. I’m shooting for around 1.3 but that’s sort of arbitrary.
Of course, before going that route, I would get my desaturase index tested and track CRP at a minimum to follow signs of inflammation.
Brad
How long do you anticipate it takes to alter human desaturate ratio with supplemental sterculia oil blunting of SCD1?
A few people reading this article on facebook were a bit confused on the DHA/EPA supplements blunting SCD1. The same with the stored Oleic acid, it is strange that your body is maintaining elevated SCD1 levels if these fats lower SCD1. Does consuming these fats differ from burning them from body fat stores?
Specifically it seems to contradict this paragraph:
“If that stored body fat has enough PUFA in it, ROS production will be insufficient to drive enough ROS production to shut down SCD1 and in fact SCD1 will be increased!”
vs.
“All unsaturated fats lower SCD1 production, polyunsaturated fat more than monounsaturated fat. This makes sense biologically. For instance, Native Americans used a lot of bear fat (much more about this in a later post, check back). Bear fat can be relatively high in the polyunsaturated linoleic acid, similar to corn-fed lard. So if a native person is eating bear grease, SCD1 will be lowered to create an overall relatively saturated blend of body fat. The same thing is true of monounsaturated fats to a lesser degree.”
Yes, this nuance has confused a lot of people and I should have been more clear. There are direct effects on SCD1 and indirect effects caused by ROS production. Things like EPA and DHA and saturated fat have small but direct effects.
Ultimately, leptin should increase the rate of fat oxidation. If your fat is sufficiently saturated, this should drive a wave of ROS (indirect), which will trump all of the direct effects.
Brad