The way that insulin decreases blood glucose is by causing the translocation of a glucose transporter called GLUT4 (GLUcose Transporter 4) from vesicles (little pockets) inside the cell to the cell membrane where it transports glucose into the cell. GLUT4 is said to be “insulin dependent”. When glucose levels inside of cells are high, glycogen synthase is activated and the glucose is stored as glycogen. Skeletal muscle is the largest tissue in the human body and is considered the primary target for insulin-stimulated glucose disposal.1 Muscle contains the largest reservoir of glycogen in the body.2 Muscle glycogen is closely regulated and influences insulin sensitivity.2
Insulin resistance of obesity is characterized by impaired skeletal muscle glucose uptake and, more specifically, by reduced glycogen storage.3
GLUT4 translocation and AMPK
A paper I referenced in the post about vitamin D activating AMPK has this intriguing graph. Notice how vitamin D regulates GLUT4 levels (“Insulin Dependent” glucose transporter in my diagram) and glucose uptake independently of insulin and that the effects of Vitamin D and insulin on glucose uptake are additive.
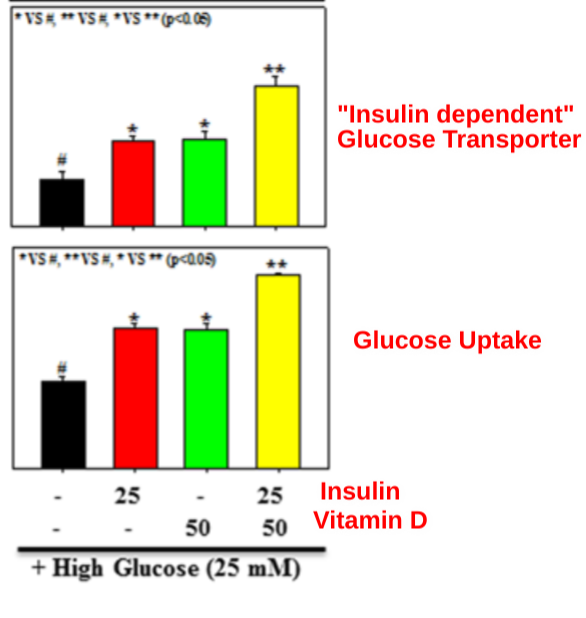
The fact that vitamin D activates AMPK and increases GLUT4 translocation independently of glucose is intriguing for a number of reasons. We’ve already seen that low SCD1 levels lead to activated AMPK. Mice lacking SCD1 have increased activation of AMPK4 and are protected from high blood glucose caused by a high fat diet5. Hampshire pigs have an overactive version of AMPK and have high levels of muscle glycogen.6 Humans with an analogous AMPK mutation to Hampshire hogs have high skeletal muscle glycogen content and low intramuscular fat.7
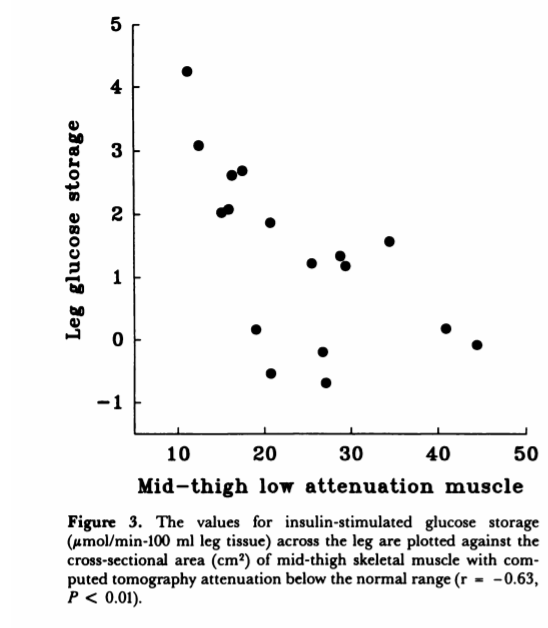
Obese humans have high levels of skeletal muscle SCD1, which is associated with high intramuscular fat levels and low levels of actiavted AMPK. High intermuscular fat levels show up as low attenuation on a CT scan. Humans with higher intramuscular fat store less glycogen.3
AMPK increases GLUT4 translocation without insulin
All of this is reason to believe that AMPK plays a large role in muscle glycogen content and blood glucose control but definitive evidence had been lacking.
Two different 2020 papers, one by an Italian group8 and one by a Korean group9, demonstrate that pAMPK increases glucose uptake, glycogen formation, and improves glucose tolerance independently of the effects of insulin. The paper from the Italian group is published in a more prestigious journal (Nature), but the Korean group looks at ROS!
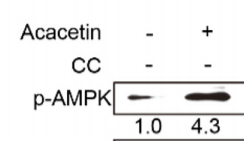
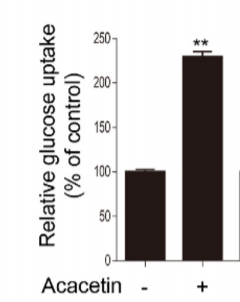
Both studies use an AMPK activator – abscisic acid (ABA) in the Italian paper and Acacetin in the Korean paper – in tissue culture and show increased phosphorylation of AMPK and increased glucose uptake due to insulin-independent GLUT4 translocation.
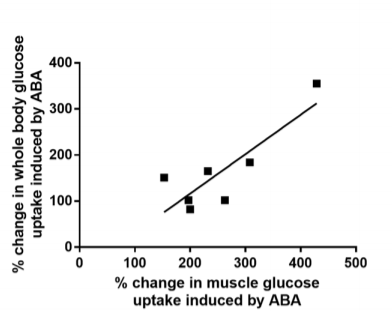
The Italian paper shows that rats given ABA have lower fasting blood glucose levels, faster whole body glucose uptake and that the rate of whole body glucose uptake is tightly correlated with muscle uptake. Muscle tissue is indeed the main sink of glucose.
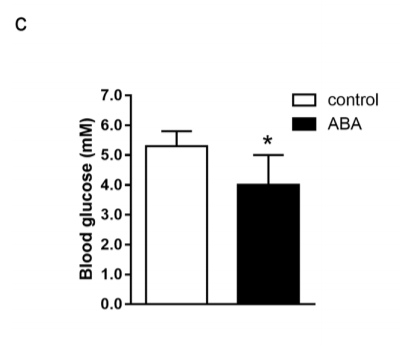
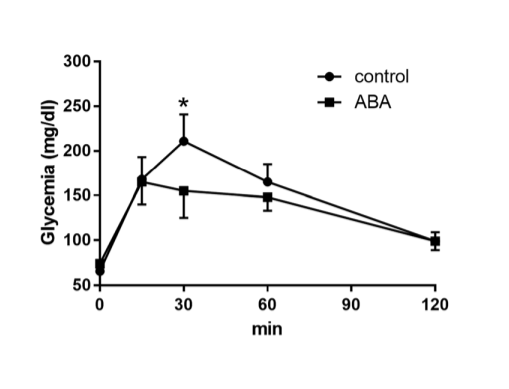
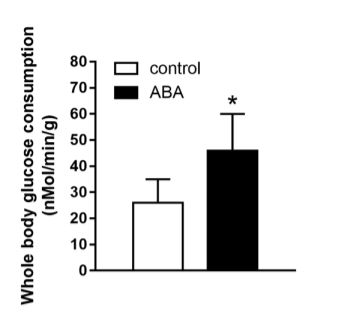
They also show that mice given ABA spontaneously spend more time running on the wheel – they are exercising BECAUSE they have pAMPK and not the other way around – and that they have a lower glycemic response to a glucose load and higher muscle glycogen content.
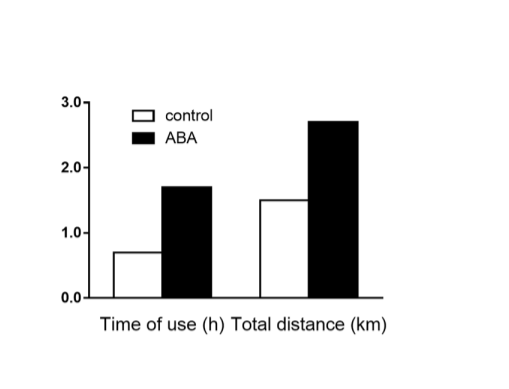
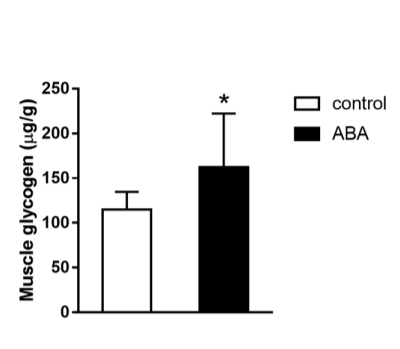
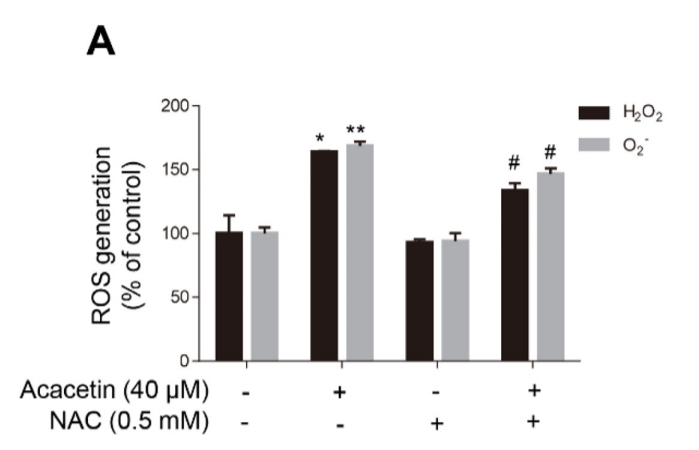
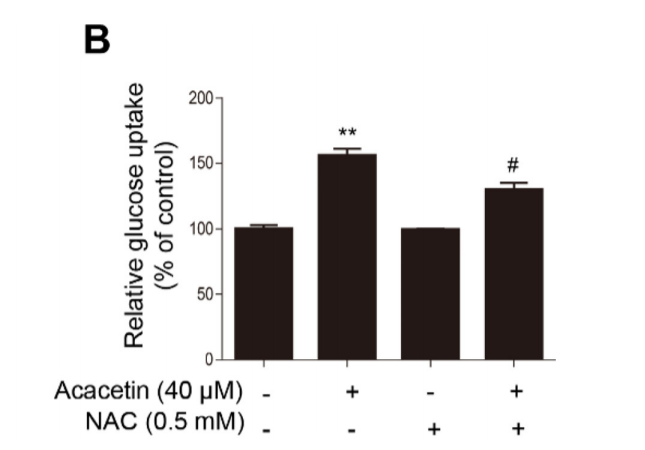
That’s all pretty nice, but the Korean paper actually looks at the role of ROS in all of this!! They show that by using NAC – an antioxidant very similar to glutathione – the amount of hydrogen peroxide and superoxide generated in the cells is reduced and so is glucose uptake. So AMPK induced GLUT4 translocation is driven (at least partially) by ROS! I wish they would have spent more time on the role of ROS, but at least they gave us something.
Conclusions
These two papers show quite clearly that phosphorylated AMPK drives GLUT4 translocation, glucose uptake into muscle tissue and glycogen formation independently of insulin signalling. Furthermore, activating AMPK in rodents leads to lower fasting blood glucose, faster clearance of glucose when given an oral glucose load and more spontaneous activity. The whole body clearance of blood glucose is driven by skeletal muscle uptake.
This increased glucose uptake is at least partially dependent on ROS production. The skeletal muscles of obese humans have high levels of SCD1 and are therefore inneficient at driving ROS. As intramuscular fat increases in obese humans, indicating higher SCD1 levels, they have lower muscle glycogen.
When you take an oral glucose tolerance test, you consume 75g of glucose and test your blood glucose 2 hours later. A value under 140 indicates normal and a value of over 200 indicates type 2 diabetes. A value between 140 and 200 is considered “insulin resistant”. But is it really insulin resistance or is it inability to generate ROS to activate AMPK to store muscle glycogen?
- 1.Deshmukh AS. Insulin-stimulated glucose uptake in healthy and insulin-resistant skeletal muscle. Hormone Molecular Biology and Clinical Investigation. Published online January 1, 2016. doi:10.1515/hmbci-2015-0041
- 2.He J, Kelley DE. Muscle glycogen content in type 2 diabetes mellitus. American Journal of Physiology-Endocrinology and Metabolism. Published online November 2004:E1002-E1007. doi:10.1152/ajpendo.00015.2004
- 3.Simoneau J, Colberg SR, Thaete FL, Kelley DE. Skeletal muscle glycolytic and oxidative enzyme capacities are determinants of insulin sensitivity and muscle composition in obese women. FASEB j. Published online February 1995:273-278. doi:10.1096/fasebj.9.2.7781930
- 4.Dziewulska A, Dobosz AM, Dobrzyn A, et al. SCD1 regulates the AMPK/SIRT1 pathway and histone acetylation through changes in adenine nucleotide metabolism in skeletal muscle. J Cell Physiol. Published online June 26, 2019:1129-1140. doi:10.1002/jcp.29026
- 5.Miyazaki M, Sampath H, Liu X, et al. Stearoyl-CoA desaturase-1 deficiency attenuates obesity and insulin resistance in leptin-resistant obese mice. Biochemical and Biophysical Research Communications. Published online March 2009:818-822. doi:10.1016/j.bbrc.2009.01.183
- 6.Scheffler TL, Park S, Gerrard DE. Lessons to learn about postmortem metabolism using the AMPKγ3R200Q mutation in the pig. Meat Science. Published online November 2011:244-250. doi:10.1016/j.meatsci.2011.04.030
- 7.Costford SR, Kavaslar N, Ahituv N, et al. Gain-of-Function R225W Mutation in Human AMPKγ3 Causing Increased Glycogen and Decreased Triglyceride in Skeletal Muscle. Groop L, ed. PLoS ONE. Published online September 19, 2007:e903. doi:10.1371/journal.pone.0000903
- 8.Magnone M, Emionite L, Guida L, et al. Insulin-independent stimulation of skeletal muscle glucose uptake by low-dose abscisic acid via AMPK activation. Sci Rep. Published online January 29, 2020. doi:10.1038/s41598-020-58206-0
- 9.Kwon E-B, Kang M-J, Ryu HW, et al. Acacetin enhances glucose uptake through insulin-independent GLUT4 translocation in L6 myotubes. Phytomedicine. Published online March 2020:153178. doi:10.1016/j.phymed.2020.153178

Cross posting here as promised in a late comment to your previous blog post:
Wait a second. I posted on twitter asking you about Stearic acid and ROS and AMPK, now I just had an epiphany-
Does Stearic acid trigger AMPK via ROS – ONLY – in the context of high glucose/starchy meal?
As explained here, a starchy meal can trigger AMPK by ROS:
—
“So when high cellular energy levels lead to ROS production, GAPDH activity stops, glucose is converted to Glyceraldehyde-3-Phosphate, which converts ATP to ADP and ATP production slows. This results in a rapid increase in the ADP to ATP ratio and AMPK is activated.
AMPK is activated due to HIGH cellular energy levels after a starchy meal.”
—
Stearic acid can increase ROS further. So can alcohol(? … not sure if it interferes with glucose though) …
So now: Anything that increases ROS in the presence of elevated glucose/starch, will trigger AMPK, and cause a massive upregulation in energy production, with combined elevated fat burning at the same time?
..which will independently upregulate GLUT4 per your latest blog post, and cause even more sugar to come into the cell, keeping the party going?
I’m trying to eat starch, rice, and even bought and scarfed down those butter croissants… coz my craving for starch had gone thru the roof…
I’ve gained 6lbs in 3 days.
But I don’t think I will be able to stay on this diet without starch.
Not sure what to do.
Any info on peeps with a screwed up carbohydrate metabolism?
thank you, sniff sniff
Brad… and Eric… both of you
My working theory so far based on Brad’s writings and personal experience is something like this-
1. PUFA and possibly other environmental factors trigger PPAR gamma through AhR, creating torpor and SCD1 which opposes AMPK even if you try to activate it. Insulin I saw on a paper’s diagram seems to oppose AMPK too.
2. AMPK can oppose torpor, but it’s a tug of war
3. The game is to minimize torporigenic factors while increasing the amount of AMPK triggers you can get
Thus:
Reducing PUFA as much as possible, any other factors that might trigger torpor (for me sugar definitely does it), and doubling down on AMPK factors – I drink hibiscus tea (4 teabags in 32oz steeped overnight in the fridge, drank throughout the day), get sunlight when I can, take sterculia oil in the morning (SCD1 inhibitor)
Combining stearic acid (meat fat from beef mostly) with starch seems to trigger AMPK and a temporary hyperactive state for me
In the past I tried croissants, they helped for a little while but eventually didn’t work for me. I believe insulin also messes with the AMPK levels (iirc from a diagram I saw in a paper). So it’s a situational thing – somehow you have to get insulin under control, sometimes that just involves traditional exercise & eating less. It’s a huge bag of factors that you’re juggling to get the tug of war in favor of AMPK and less in favor of PPAR gamma/SREBP-1c/SCD1/torpor.
Sometimes it’s just time you need – working slowly & steadily on the goal of reducing fat mass (higher fat mass and hypertrophic fat cells can leak fat into the bloodstream and cause the liver to perform inappropriate gluconeogenesis, raising blood sugar and insulin, I believe this is what pre-diabetes really is).
Anyway that’s a lot of spitballing so I apologize if it’s not super helpful but I can say have faith, really look at the different factors and how they apply to you & try to maintain course.
ok, thank you, Eric…. i am going to start drinking that tea you drink…
all i eat is rice and grass type ground beef, but i can’t live with so little variety, and the diet doesn’t seem to be working anyway…
sometimes i will go in the kitchen and read the labels on all my husband’s food and it’s impossible for me to find something without sugar/PUFAs…
i feel deprived and depressed, and like i said, this doesn’t seem to be working
i’ve got ALL the supplements, and i’m trying so hard…
sniff sniff, what with a huge case of torpor and pain, exercise? lol
thanks so much for answering! xoxoxox
This for Elaine K, but couldn’t reply to her for some reason. I recommend looking into Perfect Health Diet. It follows Brad’s thinking very closely regarding not eating PUFAs but provides a better framework for lifelong eating and reasons for eating within said framework. https://perfecthealthdiet.com – I recommend reading the book rather than starting with the website.
Elaine – I’ve found that you have to make sure you’re working to lower your SCD1 levels initially.
a good ripe pu erh tea and sterculia oil goes a long way. I’ve eaten this way both with and without having tea and sterculia oil, and I have more success when I use them.
just rice and ground beef isn’t sustainable.
steaks, stew meat, etc are all good. very lean chicken breast and pork are good too, but maybe in a little more moderation. also, try and come up with a few recipes utilizing ground beef. I have spaghetti, chili, taco, and stir fry recipes that all use ground beef and it definitely help with variety.
Have you watched David’s videos on youtube? He also gives some good examples of foods you can eat.
And I can personally attest to the deliciousness and satiety from eating a huge pancake fried in a ton of beef tallow made from grass feed beef suet (which is a high stearic acid fat) , smothered in butter with whip cream and strawberry jam lol
you can have almost any item you can think of from regular ol’ american diet, but you’ll likely have to find a recipe that you can tweak to bring in line with the principles here, and you’ll just have to make a lot of your own food or snacks.
my next goal is to create a recipe for cinnamon rolls that fits nicely into the croissant diet.
but I think what might make the biggest difference at first is the sterculia oil and pu erh tea. look for ripe, fermented, or shou pu erh, these are all the same thing and it’s the kind you want for the effect of reducing SDC1.
like I said, I’ve tried with and without, and I’ve had better progress with. from all the information here, I think using supplements to lower SCD1 might be a good idea until body fat levels are more unsaturated and the feedback loop is broken.
Also, Eric, I meant to say thank you for the encouragement to stick it out, give it time
i think i have thyroid problems too…
cheers,
Elaine xox
@Elaine
I was just thinking, from past years when I used to hang around low-carb forums, your situation sounds eerily similar to some accounts of hypothyroid problems. Usually it was women complaining of it too, I am really curious what that’s all about… I’m assuming you have a doctor helping you with this or no?
Hmm. I was finding that it worked best for me when I kept Brad’s suggested macros (recast as: no concentrated PUFAs; grams fat > grams carb; grams saturated > grams unsaturated) in mind for all the meals. I’d already been drinking puer. That, finding the remnant vegetable oils, and focusing on the spiked butter oil stopped the weight gain.
Adding Sterculia oil, berberine, and astaxanthin to that have lost most of the weight that crept on while I was finding the many ways the rest of the family insisted on keeping vegetable oil in the diet. I do need to keep carbs down, but not off. The weight loss has accelerated with the recent mentions of the AMPK agonists; the breakfast croissant has had added Tajin (a brand-name Mexican condiment of salt, mild chile, and dehydrated lime juice), turmeric, black pepper, either Tabasco or crushed red peppers, and some glycerol monostearate, all of which together are further boosting the loss.
It does sound to me like you aren’t eating enough, or widely enough, to trigger AMPK. Pulled roasts seem to have a better fat ratio than the ground beef. Daily I’ve been having the breakfast croissant as above, I may or may not have some ‘sandwiches’ of cheese slices with store-bought deli sliced roast beef (Kroger’s store brand has 2.5g fat, 1.5 saturated per 2 oz; yellow mustard has turmeric in it so add black pepper for absorption) with the noon-time berberine, and dinner is either some of what the family is having or if they want something PUFA laden, a pita spread inside with Brad’s recipe butter oil and filled with leftover pulled beef. Snacks when necessary are a couple squares from a Ghirardelli 86% chocolate bar or a half-serving of gelatin gummi bears.
Jut some suggestions that seem to be working for me at the moment….
And on the same line as making sure, before we start disassembling the lamp, that the lamp is plugged in, that the switches on the lamp and for the outlet are on, and that power is available at the house….
You _are_ cooking the rice yourself from dry, rinsing it three times or when the water stops getting cloudy, etc. and not using the precooked rice bags/cups where the grains are separated by a thin coating of vegetable oil, correct?
I didn’t realize they did that with precooked rice. Luckily we do ours from dry (not rinsing, just from a huge bag into a rice cooker w/ ghee or butter added for slickness)
The microwave precooked rices I’ve seen have the vegetable oil. The parboiled rices are usually just boiled to almost cooked, then re-dried. The rinsing is to remove the milled-off starch so the grains don’t clump together when cooked alone, not needed for parboiled (and it sounds like when cooked with the butter, either) 🙂
I tried butter croissants as well, because hey I’ll take any excuse I can get to have those. As with you my hunger skyrocketed persistently which was definitely not what I wanted to put up with, not mentally sustainable. I also gained the expected pounds of water weight from carb re-introduction, but that’s fine and can leave just as easy. Potatoes on the other hand seem to be much more sustainable, and only make me a little bit hungrier. Less “I need to eat” and more “I’m looking forward to them”. I basically make homemade hashbrowns along with my steak, they can definitely soak up a lot of tallow.
That said, I don’t think they are doing anything beneficial for me. My temperature has been a little lower, but other than that I don’t have anything like good controls or measurements going on. I’m mostly just seeing how they make me feel, which is surprisingly “normal”. We’ll see if that opinion changes when I go back on carnivore after this bag. I’ll also add that the potatoes seem to be just as good for my constipation issues as carnivore, which I had moved to from keto (meat + vegetables) for that reason.
First off, don’t panic. If you were keto before and added back starch, a quick 6 lbs may simply be glycogen repletion, AKA “water weight”. See if you stabilize.
whoa!! thanks everyone for your replies, diet suggestions, etc… and Brad, yes I was coming off of keto!
Erich, yes I’m making my own rice, and taking sterculia oil. and i’m definitely not eating enuff, i have a small stomach and my cocoa butter coffee makes me soooo full i can’t take it lol
Eric, no I’m not working with doctors, they have checked my thyroid with their standard test and always tell me there is nothing wrong with it, when it actually HURTS sometimes and i get anxiety COMING FROM the area of my thyroid, it’s ridiculous, but I’m in canada and they can’t be bothered to test me further.
Nix, normally, when I eat starch and fat it’s soooo bad, i gain tons of weight super fast and my immune system does whacky things… but with potatoes and the right croissants, i’m not gaining AS MUCH and my immune system is fine, so that gives me hope…
thanks so much everyone, sorry if my reply is lacking, i just got up lol yep, circadian rhythm probs too 🙂
Elaine xox
OK Gotcha – good to know – if they “checked your thyroid” then chances are you have enough iodine in your diet and TSH is normal. My understanding is it’s all about T4->T3 or rT3, and tbh you’re probably in a similar boat as many of the rest of us right now, albeit maybe one of the more resistant cases.
I suspect we all sit on some sort of continuum of “lightly torpid” to “severely/resistantly torpid”, and I was encouraged by some of Brad’s reports of success among his readers; I suspect some of those folks, including the dude who ate ice cream every day, were closer to the “lightly torpid” or maybe “torpid but responded very well to basic intervention” whereas I know I am a bit more “resistant” of a case, and you may be even more resistant to the interventions. What I love about Brad’s series is with every major enzyme system he talks about, we find different therapies that target them (e.g. SIRT1, from Brad’s newest post, was all the rage years back due to resveratrol being in wine and folks talking about wine; AMPK, from the recent series of blog posts, well just google that you’ll find a zillion things that trigger AMPK…)
Hi Eric… oh yes, i’m SUPER torpid and resistant
and that’s a good idea to search for the ampk activators… i duck duck go’d how animals get out of torpor and found a site that said animals get out of torpor with ‘violent shaking and muscle contractions’ for about an hour.. i figured that sounded like zumba class 🙂 but i don’t have the ability for exercise…
i’ll go and google now 🙂 thnx xoxoxox
Sneak preview: They actually get out of torpor by upregulating sirtuins. I’m sure the waking part involves some thrashing 🙂
OK, just checking to make sure it wasn’t something painfully simple. (I’ve solved too many problems by playing Captain Obvious to not check when something like that occurs to me.)
Another thing I’d noticed was at the beginning I didn’t seem to be using the sterculia oil as fast as I’d expected (1 tsp is 1/6 of an ounce, so if I wasn’t using an ounce a week, I wasn’t getting enough). A third dropperful in the morning fixed that and started the weight loss. You do want a minimum effective dose, so are you getting that much?
With Brad going over the AMPK activators, adding in what I have of those is further hitting the accelerator. (Thanks, Brad! I’m finding the bio-chem _very_ useful.)
This may be slightly off topic, but I was reading about torpor in animals and it is thought that in many of them (esp the little ones with fast metabolisms) a drop in available calories may lead to torpor also. I wonder if in humans, in addition to abundant PUFAs that sort of signal autumn/ winter and an upcoming lack of calories, an acute lack of calories (like in long-term or repeated dieting) may also push towards a lower metabolism/ core temp. I suppose a fuel partitioning disorder is a lack of calories at the cell-level, but wonder if calorie restriction is this at a macro organism level.
Have you talked about this before Brad? Sorry if I have missed it and this is redundant!
I’m actually writing an article about exactly this at the moment.