I’m using the word global to mean “in all tissues of the body” but I’d forgive you if you thought that I meant “in all countries where they eat vegetable oil”. Either way, really.
As I explained in part 1, the production of mitochondrial ROS is a thermogenic loop that wastes calories as heat. If you are a hibernating animal trying to sleep through winter, you lower your metabolic rate and body temperature to preserve fat mass. The last thing you want to do is waste calories as heat.
ROS are produced in the mitochondria when energy levels are sufficient and you have lots of electrons entering the electron transport chain through complex I and complex II at the same time. Both I and II have to hand electrons to coenzyme Q, creating a bottleneck. When the bottleneck is overwhelmed, electrons flow backwards, back into the mitochondria, creating superoxide. This wastes energy and creates heat.
Complex II is also known as succinate dehydrogenase (SDH). The electron transport chain is fed largely by the citric acid cycle, which burns acetyl-CoA – derived from both carbohydrate and fat – as fuel. Acetyl-CoA combines with oxaloacetate to form citrate. Citrate is converted to succinate, converting two molecules of (soluble) NAD+ to NADH in the process. Succinate then hands its electrons directly to succinate dehydrogenase (SDH AKA complex II), which converts it to fumarate. Fumarate converts one more NAD+ to NADH to become oxaloacetate again, restarting the cycle.
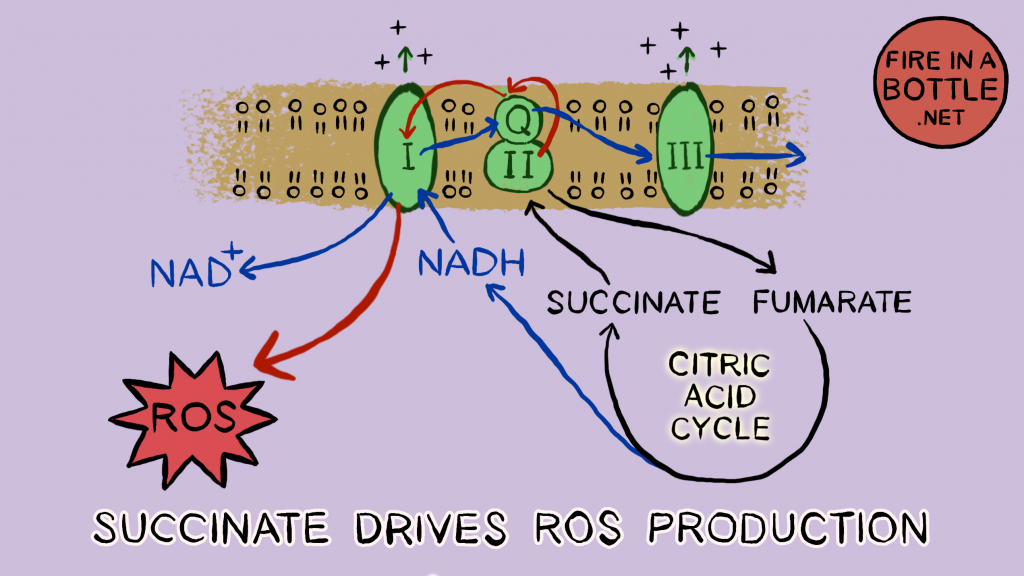
When saturated fat is metabolized, every time an acetyl-CoA is produced electrons are passed directly to coenzyme Q through acyl-CoA dehydrogenase, which behaves much like succinate dehydrogenase in producing ROS. They are both FADH2 inputs. The effects of acyl-CoA dehydrogenase and succinate dehydrogenase are additive and so burning saturated fat drives a lot of ROS, which generates heat and provides NAD+ to keep metabolism humming. For this article I am going to refer to the collective activities of succinate dehydrogenase and acyl-CoA dehydrogenase as “SDH activity”.
Linoleic acid reduces SDH activity
When a ground squirrel wants to hibernate, it eats acorns. Acorns are loaded with highly unsaturated fat – most of it is monounsaturated and 20% or so is polyunsaturated (PUFA). Squirrels who don’t eat enough PUFA fail to enter torpor. Each unsaturated bond is one less FADH2 input and lowers SDH activity. Low SDH activity means minimal ROS generation.
When lots of energy is available – the animal is gorging on acorns and has plentiful bodyfat – ROS generation is necessary to regenerate NAD+ via NNT to continue running the citric acid cycle. If ROS cannot be generated, NAD+ pools are depleted. This is reductive stress: all NAD+ – the oxidized form – have been converted to NADH, the reduced form. There are no oxidants to give NAD+ back.
When NAD+ is low SIRT3 is deactivated and SDH is turned OFF. Low SDH activity begets low SDH activity.
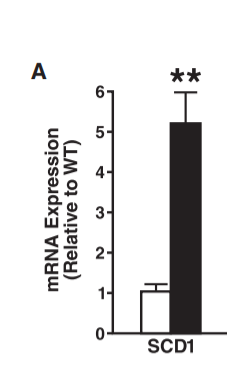
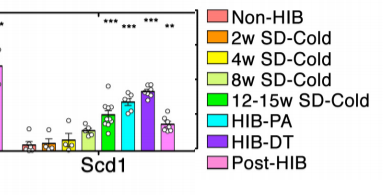
When SIRT3 is low, SCD1 levels ALSO rise, creating more unsaturated fat which lowers SDH activity. Look at how the SCD1 levels EXPLODE in a SIRT3 knockout mouse (they don’t have ANY SIRT3) on a high fat diet1. Also, SCD1 levels rise as a squirrel approaches torpor.2
Low SDH activity begets low SDH activity.
Succinate Dehydrogenase is turned off to enter torpor
Hibernating animals cycle between bouts of torpor lasting 7-14 days and much shorter times of arousal, when they rapidly increase their metabolic rate and body temperature.
SDH activity is typically 50% lower upon entry into torpor compared to when the squirrels are aroused.3
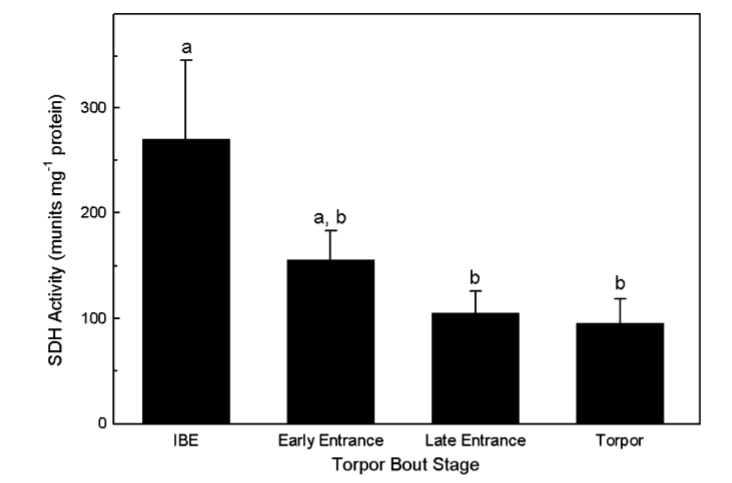
In late torpor – in the days immediately preceding arousal – when the squirrels are getting ready to rapidly increase their metabolic rate, they massively increase the activity levels of their SIRT enzymes, deacetylating SDH, allowing them to increase their metabolic rate.4
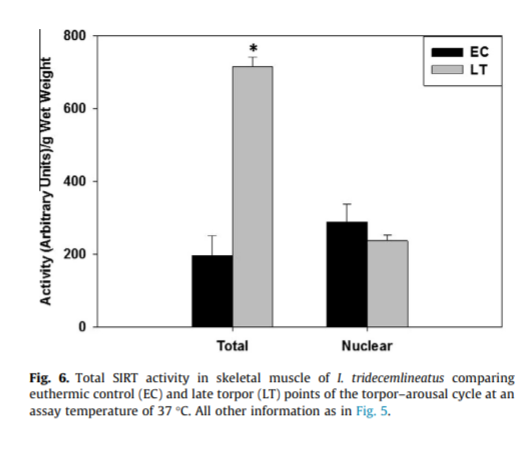
Upon arousal, SIRT levels return to normal. With highly unsaturated body fat and high SCD1 levels, NAD+ levels are quickly reduced, SIRT3 activity level drops, SDH activity plunges, metabolic rates drop and the squirrels re-enter torpor.
Obese humans have low SDH activity
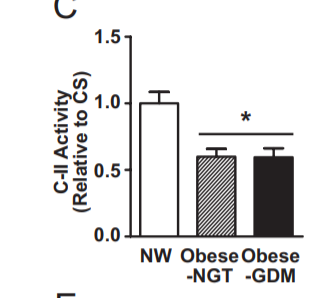
I’ve only found one paper that looks at SDH (Complex II) activity in obese versus lean actual walking-around humans. This study of pregnant women showed that obese mothers – whether or not they had gestational diabetes – were specifically low in SDH actvivty. They weren’t low in complex I activity or citrate synthase activity (which initiates the TCA by incorporating a new acetyl group from acetyl-CoA).
They were specifically low in SDH activity, which was reduced by about half. Just like in the getting-torpid squirrels. And obese humans have low metabolic rates in muscle and adipose tissue5.
The third paper
This brings me to the third paper6 that I referenced in part 1, which complements the first paper very well. The first paper showed that mice lacking SCD1 have ratios of NAD+/NADH and AMP/ATP. When those things happen together, both AMPK and the sirtuins SIRT1 and SIRT3 become activated. This activates PGC1a which leads to thermogenesis, which is marked by increased expression of the mitochondrial uncoupling protein UCP1. This paper shows that the reason things lacking SCD1 are hypermetabolic (high metabolic rate) is due to high SDH activity and high ROS production.
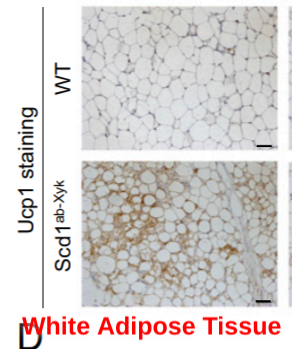
The authors did staining for the thermogenic uncoupling protein 1 (UCP1) in the white adipose tissue of mice lacking SCD1. At one point it was thought that UCP1 was specific to brown adipose tissue – the thermogenic type of fat tissue that mice have lots of and humans have a little of. These days it is known that white adipose tissue can undergo a process known as “beige-ing” where some of the white adipose cells will become more like brown adipose tissue – they will have hugely increased density of mitochondria and will express large amounts of uncoupling protein. These fat cells are there to burn energy and create heat, not to store fat.
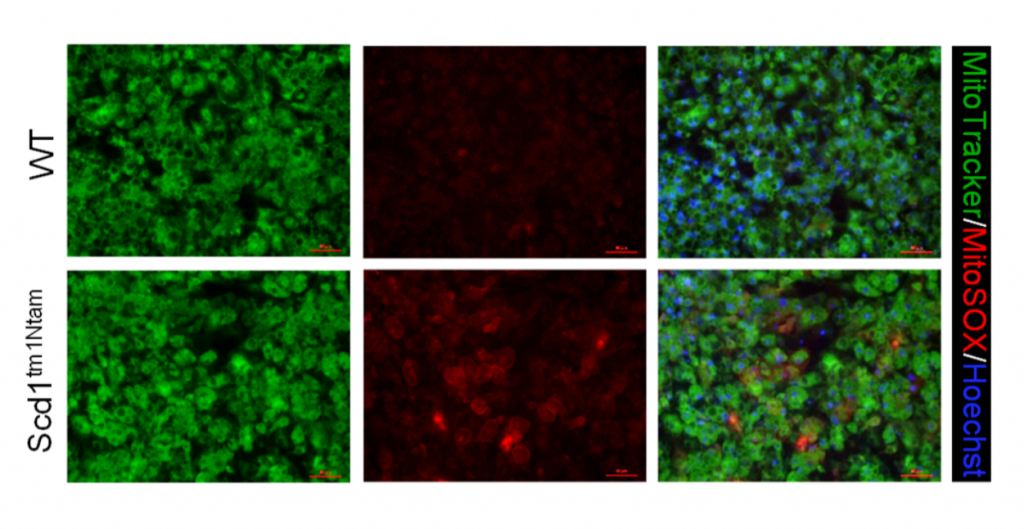
As you can see from the UCP1 staining, mice lacking SCD1 have massively enhanced numbers of beige adipocytes within their white adipose tissue. The reason they are hypermetabolic is that they are doing thermogenesis!
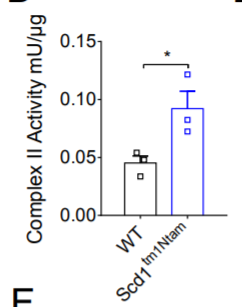
The paper shows that fat cells lacking SCD1, burning highly saturated fat, generate much more ROS than normal cells.
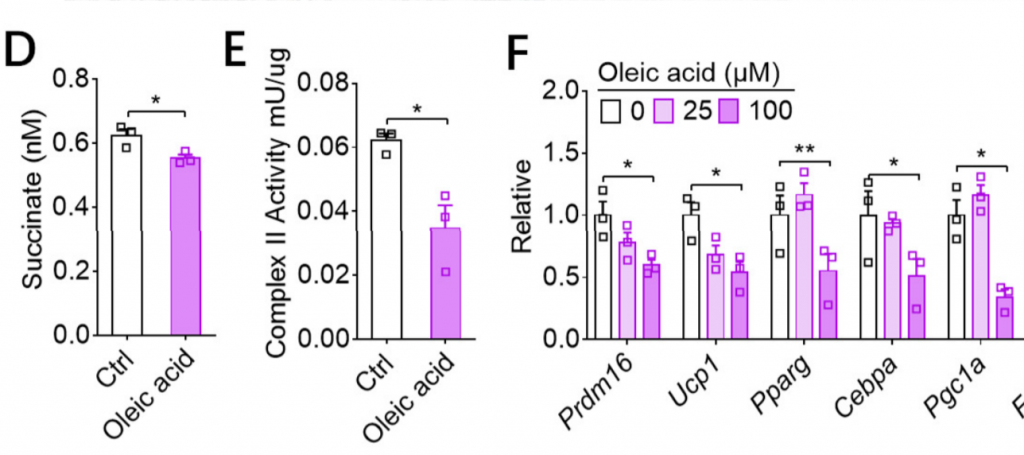
The relative levels of succinate, SDH activity and thermogenic genes in cells lacking SCD1 can all be decreased by feeding the cells oleic acid – a monounsaturated fat which reduces SDH activity.
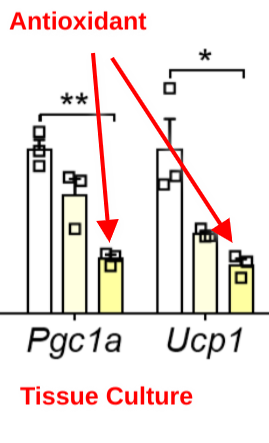
Mitoquinone – a mitochondrial antioxidant – significantly reduced the expression of thermogenic genes. ROS causes white fat cell beige-ing and thermogenesis.
The paper shows that the reason mice lacking SCD1 are hypermetabolic is that they have high SDH activity leading to high ROS formation. Paydirt for The ROS Theory of Obesity. Oleic acid and antioxidants reduce thermogenic gene expression. It is no wonder that squirrels increase SCD1 and acetylate SDH to lower their metabolic rate.
Increasing SDH activity
I was explaining the SDH activity issue to my friend Alan and I said maybe we should just eat succinate? And he said, “That’s the answer an eighth grader would come up with.” Simple. Exactly.
Succinate actually works like stearic acid, by increasing the FADH2:NADH ratio. Every time an Acetyl-CoA goes around the citric acid cycle, two NAD+ are converted to NADH as citric acid is converted to succinate. Succinate then hands electrons directly to SDH to become fumarate and one more NAD+ is converted to NADH before succinate becomes oxaloacetate, the end product of the citric acid cycle.
That is a long winded way of saying that the FADH2:NADH ratio of succinate is 1.0. Remember that stearic acid has a FADH2:NADH ratio of 0.49. Here is the list of FADH2:NADH ratios of known macronutrients.
| FADH2/NADH Ratio | |
| Succinate | 1.0 |
| Glucose | Up to 0.5 |
| Stearic Acid | 0.49 |
| Oleic Acid | 0.46 |
| Linoleic Acid | 0.43 |
Feeding studies
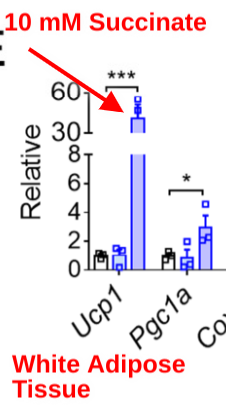
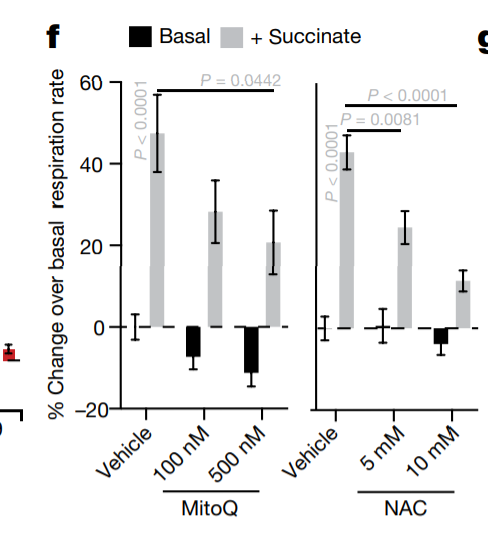
The authors actually fed regular mice a little succinate (10 mM) in their drinking water. This led to a massive 40-fold increase in UCP1 in the white adipose tissue of the mice! In addition, whole body energy expenditure increased.
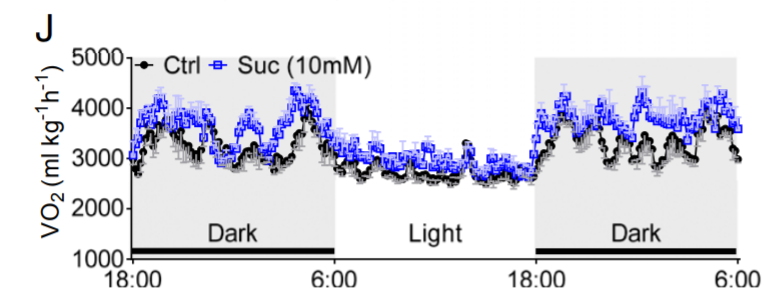
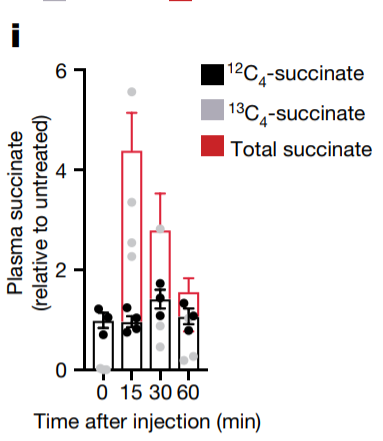
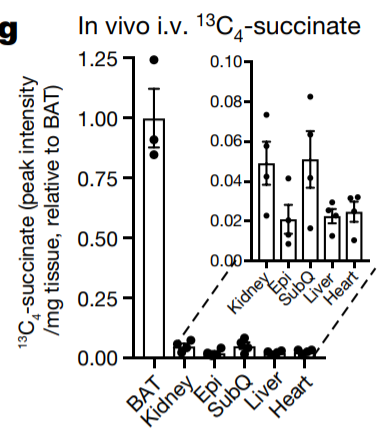
I had a lot of questions about the behavior of succinate. How quickly is it taken up from blood? Which tissues take it up? Does it make it into the mitochondria? This paper7 used radiolabelled, injected succinate shows that an increase in succinate was cleared from the bloodstream within an hour. Succinate is HIGHLY preferentially taken up by brown adipose tissue, but other tissues also take it up. My assumption is that in a human – with a lower proportion of brown adipose tissue, more succinate is taken up by other tissues.
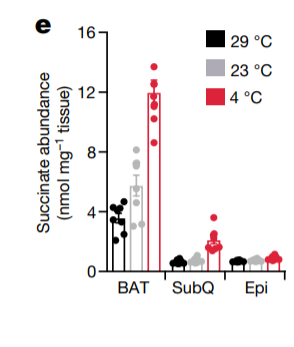
Succinate accumulates in both brown and white adipose tissue. The metabolic rate (OCR Oxygen Consumption Rate) of brown fat cells increases if you feed them succinate.
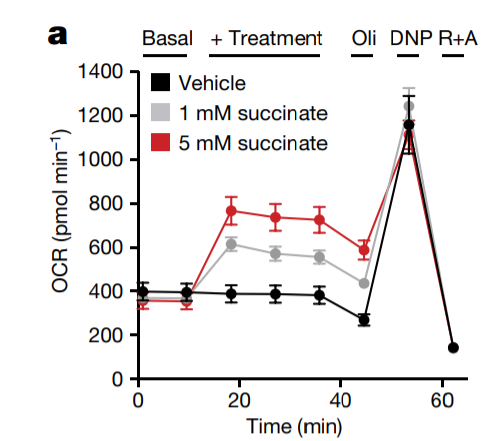
If you take the brown adipose fat cells that are cultured with succinate and you add antioxidants – either MitoQ or NAC – their metabolic rate drops in a dose dependent fashion. This is because metabolic rate is controlled by ROS generation via SDH activity which regenerates NAD+. Antioxidants reduce ROS. The black bars are not given succinate.
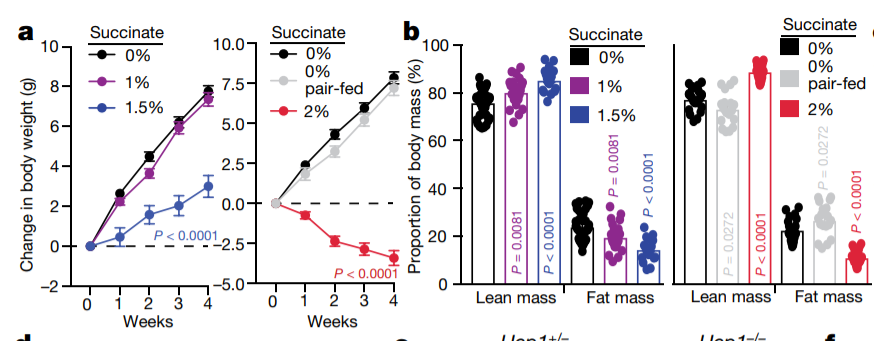
The authors ran two different experiments looking at weight gain on a “high fat” diet using dietary succinate in normal mice. The first trial used 1% and 1.5% succinate (purple and blue) and the second experiment used 2% (red). In humans, 2% would be 10g out of a 2000 calorie diet. 1.5% succinate prevented weight gain on the high fat diet and 2% produced weight loss while on the high fat diet. Mice fed succinate had higher lean mass and lower body fat. The light grey line in the second experiment are “pair fed” mice. This means that their calories were matched to the mice who received 2% succinate. The weight loss is not because of caloric restriction.
Another study8 looked at the effects of dietary succinate on muscle fiber types. Obese humans have a low percentage of oxidative type I and type IIa muscle fibers and higher amounts of the glycolytic type IIb fibers9. Mice fed 0.5% (pink) or 1% (red) succinate showed large increases in the percentage of type I and type IIa and a decrease in type IIb fibers. The succinate induced a change in muscle fiber types to one resembling a lean person.
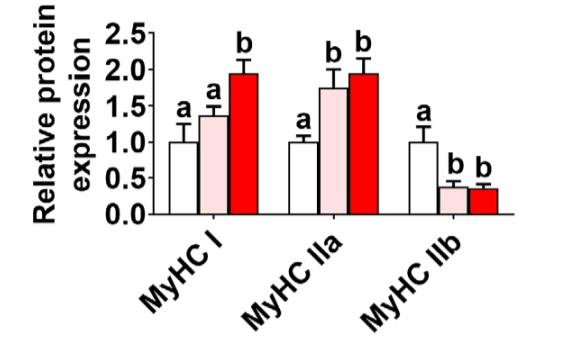
The succinate also increased grip strength and endurance in the mice.
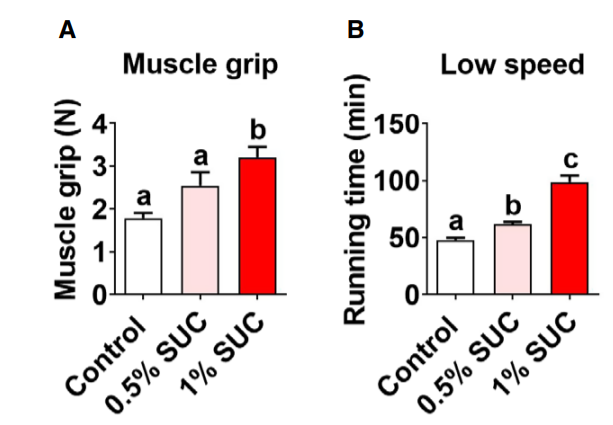
The succinate also increased SDH activity and metabolic rate.
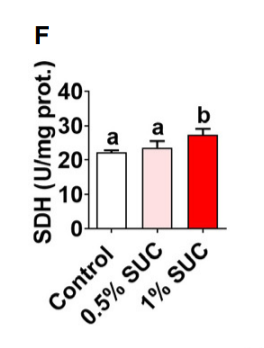
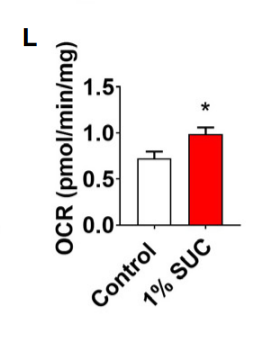
Summary of succinate benefits
OK, so let’s add it up. In rodent models, succinate is absorbed very preferentially by brown adipose tissue where it induces thermogenesis. It also induces stem cells in white adipocytes to become beige adipocytes, again leading to thermogenesis. IIt directly increases metabolic rate. It increases lean mass, reduces fat mass and causes weight loss. It also causes fiber type conversion of skeletal muscle from the glycolytic type seen in obesity to the oxidative type leading to increased strength and stamina.
Anything else? It increases blood glucose control in mice fed a high-fat, high-sugar diet10 and it increases tight junction protein in pigs11, creating better intestinal barrier function. Also, gut bacteria generate a lot of succinate from fiber10 So it may mimic the beneficial effects of dietary fiber.
Many of these benefits are caused directly by succinates ability to drive ROS production via SDH. We know this since decreasing ROS production – either by using an antioxidant or by supplementing with unsaturated fat – reduces the thermogenic conversion of white adipocytes and reduces the increase in metabolic rate due to succinate supplementation.
Are there downsides?
YES! It can be quite laxative. I suspect this is due to ROS production in the intestinal cells, but that’s just me guessing. If you choose to supplement with it, take it easy at first, see how it goes.
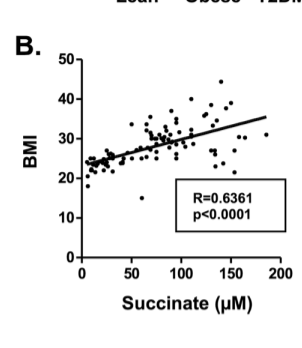
There is also the fact that circulating succinate levels are actually increased in obesity and diabetes. I attribute this to the fact that obese people and diabetics have low succinate dehydrogenase activity, so succinate builds up in cells and is exported, leading to an increase in blood levels. Presumably it is ultimately taken up by brown or beige adipose tissue when blood levels get high enough.
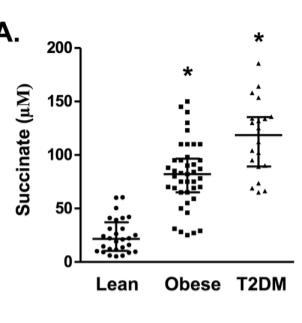
Lastly, succinate has been shown to be associated with inflammation in some cases. It’s not always clear whether this is cause or effect, and results have been contradictory. This paper12 showed that succinate triggers macrophage activation through the succinate receptor SUCNR1. This paper13 showed that succinate suppresses immune response via a SUCNR1 independent pathway. This is definitely not a settled matter. My thoughts are to take it slow and see what happens. If you have inflammatory bowel disease or Chrons, succinate may not be the right choice for you.
A-lipoic acid, succinate’s little buddy
You’ve probably had the thought, “What is the point of eating succinate if my succinate dehydrogenase is acetylated anyway?” That is where a-lipoic acid comes in. In the past I’ve recommended resveratrol for activating SIRT1 to get things de-acetylated and moving. There is another polyphenol that is very good for SIRT1 activation called myricetin (spelled like ‘my rice tin’, thank you auto-correct for the tip). But the best thing for this IMO is a-lipoic acid (“alpha lipoic”).
A-lipoic acid is made by mammals and found in relatively high amounts in the liver, so it is a natural substance of humans. It seems to be relatively absorbed on an empty stomach and is taken up in cells within an hour or so14, where it is reduced to dihydrolipoic acid in a reaction that converts NADH to NAD+, after which the dihydrolipoic acid is transported out of cells and eliminated in urine. Which is to say that if you take a-lipoic acid as a supplement you can directly unload reductive stress, regenerate NAD+ and activate SIRT3!
A-lipoic acid is sold as an “anti-oxidant”, but you actually take it in the oxidized form. When you reduce it to dihydrolipoic acid, you oxidize NADH to NAD+. So a-lipoic acid allows things to be oxidized. It is a source of oxidant power. It eliminates reductive stress. Here’s the great part, though… Once you’ve reduced it, it actually DOES function as an antioxidant, eliminating a superoxide molecule, which then allows another molecule of NADH to become NAD+ again. A-lipoic acid and dihydrolipoic acid act as an NAD+ regenerating redox couple.
It is also incredibly safe15, showing no side effects in rats of a dose that would be equivalent to 6000mg in adult humans. Lastly, a-lipoic acid supplements are cheap and readily available. The R-alpha-lipoic acid is the one preferred in the mitochondria.
In fact, the story of a-lipoic acid is so interesting that it will get its own article. Next year, if I have time…
Succin-ade
So I got some succinate to try it. WOW! It’s VERY sour!!! Punishingly sour. It IS part of the citric acid cycle, after all. Have you ever tried to eat just straight citric acid? Citric acid is what they mix with sugar to provide the sour flavor in Sour Patch Kids. It’s also has an odd bitter flavor. I thought about getting capsules made, but the amounts shown to produce weight loss in studies – 1-2% of diet – suggested using something like 5-15g of succinate per day. At 800mg that’s a lot of pills to swallow!
There is also a di-sodium succinate. The sodium removes the sour flavor (but not the bitter flavor) and makes it much more water soluble! Unfortunately it’s quite expensive. On the other hand you don’t need THAT much.
I found that by tinkering with the proportion of di-sodium and regular succinate, adding a little monkfruit extract and natural lemon-lime flavor, I was able to come up with a powdered mix that tastes something like lemonade. Sour from the succinate and a bit salty like a margarita. The sweetness of the monkfruit mostly hides the bitterness and a scoop (5g of succinate) costs about a buck. It’s not great but neither is it horrible. My friend said it tastes like “lemonade for astronauts”. That’s about right. You could add it to a stearic acid smoothie if you were inclined.
For non-US customers, I am offering free international shipping with the purchase of four items selected from Succin-ade, sterculia oil and stearic acid (limited to two stearic acid which is in short supply at the moment).
A human n=1 trial of Succin-ade
A friend approached me who was interested in losing weight. Other approaches she tried had failed. We met for coffee and gave her a container of Succin-ade. She was already gluten free and avoided most carbohydrates. She got a high percentage of calories from ethanol (she’s a friend of mine, like I was saying…).
I told her to avoid sources of vegetable oil, olive oil, nuts and peanut butter and to take a scoop or two of the Succin-ade every day. I told her not to worry about calories. I told her to get a scale, a sewing tape measure and a digital thermometer that takes 60 seconds to record your body temperature. The quick read ones are inaccurate. I told her to take the three measurements each day and she said she loved spreadsheets. Perfect.
She told me over the course of the experiment that she woke up, peed and then took all three measurements every day. In one month she lost 12 lbs, four inches from her waistline and her body temperature jumped from 97.1 to an average of around 98.6 (sometimes higher). She’s doing thermogenesis!
She did lose 5 lbs the first day, which is probably just random fluctuation, but she continued to lose pounds and inches. I had told her to try resveratrol and myricetin because I hadn’t realized the full potential of a-lipoic acid at the beginning of the trial. If the trial started today I would have told her to take a high dose of a-lipoic acid in the morning while fasted. Perhaps in the range of 1200-2400mg.
Then I would’ve told her to drink the succin-ade 30-60 minutes before meals, with the idea that cells will preload with succinate will essentially up-saturate the food. By my math 5g of succinate is the equivalent of swapping 12.4 of oleic acid out with stearic acid.
These are the complete results of her trial.
| Day | Weight Change | Waist Change | Body Temp | |
| 0 | 0 | 0 | 97.1 | |
| 1 | -5 | 0 | 97.1 | |
| 2 | -5 | 0 | 97.1 | |
| 3 | -6 | 0 | 96.9 | |
| 4 | -8 | -0.5 | 96.9 | |
| 5 | -6 | 0 | 97.1 | Added Vit D & K2 |
| 6 | -6 | -0.5 | 97.1 | |
| 7 | -9 | -1 | 97.1 | |
| 8 | -9 | -0.5 | 97.1 | |
| 9 | -8 | 0 | 97.6 | |
| 10 | -10 | -0.5 | 97.9 | |
| 11 | -10 | -1 | 99.1 | |
| 12 | -8 | 0 | 97.9 | |
| 13 | -6 | 0 | 97.9 | Added Resveratorl + Myricetin |
| 14 | -8 | -0.5 | 98.1 | |
| 15 | -8 | -1 | 97.9 | |
| 16 | -8 | -1 | 98.1 | |
| 17 | -10 | -1.5 | 98.1 | |
| 18 | -11 | -2 | 98.4 | Took a break |
| 19 | -11 | -2 | 98.6 | Took a break |
| 20 | -15 | -2 | 98.6 | Restarted Succin-ade |
| 21 | -13 | -3 | 99.1 | |
| 22 | -13 | -3 | 99.1 | |
| 23 | -13 | -2 | 99.1 | |
| 24 | -12 | -3 | 99.6 | |
| 25 | -12 | -3 | 99.6 | |
| 26 | -13 | -4 | 98.6 | |
| 27 | -13 | -4 | 99.1 | |
| 28 | -12 | -4 | 98.4 | |
| 29 | -12 | -4 | 99.1 |
Conclusions
Mitochondrial ROS production eliminates the reductive stress brought about by consuming unsaturated fats and overexpressing SCD1. Mitochondrial ROS is primarily driven by succinate dehydrogenase (SDH) activity. Obese humans and torpid mammals have very low SDH activity due to both highly unsaturated body fat and due to the fact that SDH is acetylated and turned OFF.
We can activate SIRT3 to deacetylate SDH with a-lipoic acid. This opens the door to supplementary succinate, which really ramps up ROS production to eliminate reductive stress and increase metabolic rate.
- 1.Hirschey MD, Shimazu T, Jing E, et al. SIRT3 Deficiency and Mitochondrial Protein Hyperacetylation Accelerate the Development of the Metabolic Syndrome. Molecular Cell. Published online October 2011:177-190. doi:10.1016/j.molcel.2011.07.019
- 2.Chayama Y, Ando L, Sato Y, et al. Molecular Basis of White Adipose Tissue Remodeling That Precedes and Coincides With Hibernation in the Syrian Hamster, a Food-Storing Hibernator. Front Physiol. Published online January 28, 2019. doi:10.3389/fphys.2018.01973
- 3.Chung D, Lloyd GP, Thomas RH, Guglielmo CG, Staples JF. Mitochondrial respiration and succinate dehydrogenase are suppressed early during entrance into a hibernation bout, but membrane remodeling is only transient. J Comp Physiol B. Published online January 5, 2011:699-711. doi:10.1007/s00360-010-0547-x
- 4.Rouble AN, Storey KB. Characterization of the SIRT family of NAD+-dependent protein deacetylases in the context of a mammalian model of hibernation, the thirteen-lined ground squirrel. Cryobiology. Published online October 2015:334-343. doi:10.1016/j.cryobiol.2015.08.009
- 5.Boyle KE, Newsom SA, Janssen RC, Lappas M, Friedman JE. Skeletal Muscle MnSOD, Mitochondrial Complex II, and SIRT3 Enzyme Activities Are Decreased in Maternal Obesity During Human Pregnancy and Gestational Diabetes Mellitus. The Journal of Clinical Endocrinology & Metabolism. Published online October 2013:E1601-E1609. doi:10.1210/jc.2013-1943
- 6.Liu K, Lin L, Li Q, et al. Scd1 controls de novo beige fat biogenesis through succinate-dependent regulation of mitochondrial complex II. Proc Natl Acad Sci USA. Published online January 17, 2020:2462-2472. doi:10.1073/pnas.1914553117
- 7.Mills EL, Pierce KA, Jedrychowski MP, et al. Accumulation of succinate controls activation of adipose tissue thermogenesis. Nature. Published online July 18, 2018:102-106. doi:10.1038/s41586-018-0353-2
- 8.Wang T, Xu Y, Yuan Y, et al. Succinate induces skeletal muscle fiber remodeling via SUCNR1 signaling. EMBO Rep. Published online July 18, 2019. doi:10.15252/embr.201947892
- 9.Tanner CJ, Barakat HA, Dohm GL, et al. Muscle fiber type is associated with obesity and weight loss. American Journal of Physiology-Endocrinology and Metabolism. Published online June 1, 2002:E1191-E1196. doi:10.1152/ajpendo.00416.2001
- 10.De Vadder F, Kovatcheva-Datchary P, Zitoun C, Duchampt A, Bäckhed F, Mithieux G. Microbiota-Produced Succinate Improves Glucose Homeostasis via Intestinal Gluconeogenesis. Cell Metabolism. Published online July 2016:151-157. doi:10.1016/j.cmet.2016.06.013
- 11.Li, Mao, Zhang, Yu, Zhu. Succinate Modulates Intestinal Barrier Function and Inflammation Response in Pigs. Biomolecules. Published online September 13, 2019:486. doi:10.3390/biom9090486
- 12.Macias-Ceja DC, Ortiz-Masiá D, Salvador P, et al. Succinate receptor mediates intestinal inflammation and fibrosis. Mucosal Immunol. Published online October 2, 2018:178-187. doi:10.1038/s41385-018-0087-3
- 13.Connors J, Dawe N, Van Limbergen J. The Role of Succinate in the Regulation of Intestinal Inflammation. Nutrients. Published online December 22, 2018:25. doi:10.3390/nu11010025
- 14.Yadav V, Marracci GH, Munar MY, et al. Pharmacokinetic study of lipoic acid in multiple sclerosis: comparing mice and human pharmacokinetic parameters. Mult Scler. Published online February 11, 2010:387-397. doi:10.1177/1352458509359722
- 15.Cremer DR, Rabeler R, Roberts A, Lynch B. Safety evaluation of α-lipoic acid (ALA). Regulatory Toxicology and Pharmacology. Published online October 2006:29-41. doi:10.1016/j.yrtph.2006.06.004

I wonder if I could use this on my husky bulldog?
Also-do you still use the oil you made, or does this replace it?
Looking forward to trying this.
Hi Brad, do you have a method of shipping to Nz presently? USPS has stopped shipping here I believe. Thanks
“that mice lacking SCD1 have ratios of NAD+/NADH and AMP/ATP.”
should be “..have high ratios of NAD+/NADH..”?
(still reading the rest of this article with bated breath!!!)
Will this be helpful to someone who has a just found out their HA1c is 6.2 ( guess pre-diabetic)
Not needing to lose weight. Slim
Not on any meds
Age 59
I’ve ordered your Butteroil in the past. Thinking about trying the “Succinate” powder drink, sounds interesting.
Thanks
Blood glucose seems intimately related to levels of pAMPK and SIRT1/3, so it may indeed have some utility here but time will tell.
Looking at the R-alpha-lipoic acid form of alpha-lipoic acid for sale, it’s is quite expensive.
Here in the UK your recommended dose of 1200mg daily would cost me £100-175/month.
Is using this R form important? I see some marketing pages claiming it is, but it’s about 10x more expensive.
I also see supplements with “d-Biotin”. What is the significance of this? Anything we should care about?
My suggested dosing is indeed quite high! I may lower it. I’m looking at “regular” a-lipoic (both types together) and it is 240 600mg capsules for 35 bucks. This seems very well absorbed, so I’m not sure it’s a case where it makes sense to spend a lot more for better absorbed formulations.
Dang. Ordering some succin-ade & a bottle of 600mg R-Alpha Lipoic Acid to test. So interesting to try this out. So basically if there was a way your body could “hydrogenate” MUFAs, the net effect would be similar to what this combination does.
PS- Would this work fine as a supplement while fasting? I can’t see why not, take R-ALA and succin-ade and…. continue to ping complex II while you’re busy burning oleic acid-rich body fat. Or is there something else happening in fasting that acetylates the mitochondrial enzymes?
I had a glass of the succin-ade while fasted. A few hours later I went to eat my steak, after 1-2 bites I pushed the plate away from myself, so incredibly full that I couldn’t bear to look at it. Then minutes later I was at the sink throwing up my 2 bites and retching until there was nothing left. I’ve since tried it with food, seems to work better, but I’m still unable to have a whole glass without intense nausea. I split one glass over 2 days with food. No weight loss yet, but my weight is steady even as I’ve indulged in ice cream, cookies, etc.
Wow, that’s a pretty intense reaction. You think the nausea was from super-satiation?
I’m halfway through my first glass of it and I’ve got some light nausea. It feels like too-much-salt nausea to me, and that makes some sense with the amount of sodium in it. Granted I have not had the reaction Kristen did.
It would be less controlled/consistent but as a food additive disodium succinate could be pretty good way add savor/salt to a dish while also getting the succinate. But could be a good, albeit pricey, way to supplement smaller amounts of it during cooking. Not sure what temps succinate breaks down at, but I’d bet its above boiling water temps.
I increase my SIRT3 with a mixture of Campari and Jagermeister about 1:1. sometime Campari with sweet vermouth.
Have a bottle of Fernet-anarca. That stuff is viciously bitter but after while it is something I reach for a “shot”.
Love Fernet – as you say it’s got strange addictive qualities.
Now I’m feeling like I need to make an Industry Sour.. ..for health and science, of course. Wonder if there’s a cocktail I can make with Campari, Fernet Branca and Green Chartreuse — that HAS to be healthy and good for you.
Is this something that must be continued?
In theory, if you can purge your unsaturated body fat and reset your enzyme systems, it could be discontinued. Time will tell if that’s possible.
This was my question too. I guess time will tell.
I get mine on Monday (15 November), so I’ll see what happens. Of course, Thanksgiving won’t help (I go off keto/low carb for at least some of that time, which means 5-7+ pounds of water weight will be packed on).
Thank you! Have two bags on order and looking forward to giving it a try. Body temp is normal around 97.4. Keep close track of sleep, HRV etc so it will be fun seeing what changes this brings about.
Let us know how it goes!
Found out about SDHI’s on twitter. Fascinating and I wonder how widespread this is- or if there are food vectors for us to ingest them (or if it even affects humans’ complex II)-
https://ag.umass.edu/turf/management-updates-apr-13-2020
Brad – surely you did your own n=1 with succin-ade – anything else to share? Inquiring minds want to know…
I did! Haha! I did an interview with Dave Tillman (to be released soon on the podcast) soon after I started. It was a hot August night and I was burning up! I had a towel to wipe the sweat off of my face but I was feeling awkward about sweating so much… At one point in the podcast I actually apologized for it!
Anyway, I experienced normalized body temp, lost 7-8 lbs and an inch on my waist but then I went on a trip and I haven’t been in my routine since. Sandra is a much better guinea pig than I! I’m planning a thorough n=1 this fall or winter using stearic acid and succinate smoothies with sterculia oil.
What an amazing research!!
Could you use succinic acid the same way? If I understand correctly, in living organisms, succinic acid takes the form of an anion = succinate?
Yeah, they’re basically different names for the same thing.
You recommend taking alpha-lipoic acid at 1200–2400 mg a day. Also stated, “The R-alpha-lipoic acid is the one preferred in the mitochondria.”
1200-2400 mg a day of alpha-lipoic acid is easy to obtain. That much of R-alpha-lipoic acid is much more expensive. Do you recommend taking the latter as opposed to the former? If so, what dosage do you recommend?
Thanks!
Ha! I should probably reduce that number. It seems pretty safe and in this case I suspect more is better. I’m honestly not sure if it matters whether it’s the R form.
What is your take on this paper?
https://www.nature.com/articles/s41586-021-04049-2
They claim that a low fat low sugar diet slows tumor growth because low sugar inhibits SCD1 so when they synthesize stearic acid they can’t convert it to oleic acid and so have unbalanced membranes and can’t grow.
On same point, how much sugar does it take to inhibit SCD1 significantly? I love fruit! How much can I eat safety?
To clarify your point, sugar increases SCD1 which is likely bad. So drinking big gulps is probably a bad idea. Can you get away with eating some fruit? You probably can. I consume Italian Amari due to the bile acid receptor stimulating effects, but they are also quite sugary. Use your judgement.
Would you please discuss Peter’s comment on Twitter?
https://twitter.com/Peter_InNorfolk/status/1454170595097301000
I’m not an expert on methyl succinate, but thanks to Eric for finding this paper: “Previous work demonstrated that methyl esters of succinate are potent insulin secretagogues in pancreatic islets, while unesterified succinate is not.”
How much R-alpha-lipoic acid do you recommend? Also, does this combo replace the stearic acid and berberine combo, or is it additional? Thanks. I’m getting back on this after being sidelined before. I was definitely seeing results too. Hopefully, this will be even better.
We are all learning together. I’ve personally dropped the polyphenols, but it’s kind of arbitrary. I’m going to trial lipoic acid, succinate, stearic and lo-dose sterculia.
I’m very curious if you have any thoughts about red light therapy (low level laser therapy), as that increases ROS and ATP production (and releases nitric oxide as a bonus). If the goal of transitioning out of topor requires higher ROS, would red light be an effective method of achieving that? I’ve used red light daily for about a year and have felt a significant increase in energy levels.
It’s not something I know much about!
Battlemouse on r/saturatedfat posted this the other day and got me curious!
https://www.reddit.com/r/SaturatedFat/comments/qk0bgc/lllt_and_atp_metabolism_thermogenesisros/
You might see my posts on twitter at some point, but if not, here’s the redundant version-
TL;DR Certain red & IR light wavelengths may resonate with the iron (heme) and copper in Complex IV, cytochrome C oxidase, and I guess (via photon-mediated pushing an electron to a higher orbital & energy state, resulting in a release of energy when the electron pops back down to its original orbital) fucks with the redox state of those carrier molecules/ions when they’re establishing the “hydroxide bridge”, potentially causing a hydroxyl radical to be released instead of OH-HO combining with 2 more protons to form two H2O’s as complex IV usually does.
Somehow this results in a Nitric Oxide being released. Not sure how/why or if it has nothing to do with iron/Cu or what.
Note that my description of iron or copper being involved is purely speculative (but reasonable educated guess IMO) – however it’s well established that Complex IV is indeed the target of this effect.
Cite: https://www.ncbi.nlm.nih.gov/labs/pmc/articles/PMC3288797/
“The relevant chromophore can be identified by matching the action spectra for the biological response to light in the NIR range to the absorption spectra of the four membrane-bound complexes identified in mitochondria.42 This procedure indicates that complex IV, also known as cytochrome c oxidase (CCO), is the crucial chromophore in the cellular response to LLLT.”
Look down towards Figure 3 for the in-depth discussion of this.
—-
Anyway, that is freaking cool. Vitamin D isn’t the only way sunlight can oppose torpor. And this has nothing to do with ultraviolet, in fact, the IR wavelength is “near-IR”, the intense heat you feel coming off a red hot cooking surface, oven, or a campfire. Near-IR blackbody radiation wavelengths dominate at >800F temps.
PS,
Curious about your red light daily routine… can you offer any details about wavelength, dose rates, coverage area, etc?
Three years ago my wife was diagnosed with macular degeneration (both wet and dry), which leads to loss of vision. Traditional docs have basically nothing to offer for dry, and the wet involves injections into the eye of anti-VEGF drugs.
I stumbled on a paper out of Germany about treatment of dry AMD with red light (740mm, just barely into the infrared). My wife’s doctor just blew me off, wouldn’t even read the paper. But as an engineer I had no trouble building a jig to duplicate the paper’s dose rates. It seems to have worked (n=2 eyes): after treating her, the doc (without knowing what we’d done) declared her eyes looked like a miracle had occurred (his words). The dry AMD was dramatically reduced.
Further study leads me to believe the effect is due to just what you alluded to: increase ATP in the RPE (retinal pigmented endothelium), and increase oxygenation of the retina due to the vasodilation effects of nitric oxide.
Anyway, sorry for the tangent, but our apparent success makes me interested in your red light therapy routine.
Hi I’m wondering if you are recommending gluten free/low carb alongside the succinade and ala or doesn’t matter? Thanks.
I think this will depend very much on the individual. Certainly many cultures are metabolically healthy on a very high carb diet/lo-fat. These diets tend to be based on white rice or tubers. The French were metabolically healthy on baguettes, wine and cheese.
Have ordered! Will try to replicate the 30 day experiment and share results on Reddit – have told ppl that I’ll be on the ‘succ-it-and-ghee’ diet 😉
Love it! Let us know!
Not really related to this article, but I read some stuff about the thyroid-axis and found this paper.
https://cellandbioscience.biomedcentral.com/articles/10.1186/2045-3701-4-38
I thought it may be interesting for you (in short – hypothyroid mice have more MUFA in their liver but not a higher triglyceride-content, treatment with thyroid-hormones increases SFAs, “suggesting the activity of stearoyl-CoA desaturase-1 was suppressed.”).
It is probably all connected.
I am concerned about the (possible) inflammation aspect of this. I plan to try it, but I want to take some inflammation markers beforehand and during:
GGT
ferritin
HS-CRP
Lp-PLA2
Anything else?
Ownyourlabs.com offers IL-6 and TNF-alpha. They’re not cheap and I felt like they mostly moved in parallel with CRP.
I priced out the ones I wrote, and they are $100 total from ownyourownlabs. So, maybe I start with these, one set of tests beforehand, and then another 1-2 weeks later.
I currently have one value for GGT and ferritin, and multiple values for CRP, so I would have at least two values for each as a starting point. Never have had Lp-PLA2 done, so that would be a new one.
I do hate having so few samples, but I can’t afford to do much more than this. One set beforehand, 2 during. And, I need to get the time to actually get to a labs place.
Yeah, this is complicated for my by the fact that I can’t use OYL in NY, so I have to drive a five hour round trip to PA for the test.
Brad – Would be good to hear more from you about the safety of SDH. . Something about it feels a bit unnatural and weird, not sure what. Good know more about the safety of it and any experiences you have had with it personally. How long did you take it for.? Did it work for you and what, if any, side effects did you observe? Have you road tested it personally to the same extent that you did with Sterculia Oil?
To be clear, SDH is an enzyme that we all have. Succinate is a metabolite of the citric acid cycle. So every time you burn fat or glucose, you are making succinate. In obese humans, the SDH activity is low in most tissues and so the succinate gets exported and blood levels rise. The succinate is presumably taken up by thermogenic fat cells – brown and beige adipocytes – and used for thermogensis. So what we are doing is subtley shifting the ratios of a natural process. Succinate is also absorbed by your gut from gut bacteria, so we take in external succinate in addition to what we make.
I am planning on doing very thorough testing of this in the next months as time and money allow. I know that others are planning on testing and posting theor results in forums such as r/SaturatedFat
Brad – Would be good to hear more from you about the safety of SDH. . Something about it feels a bit unnatural and weird. Good know more about the safety of it and any experiences you have had with it personally. How long did you take it for.? Did it work for you and what, if any, side effects did you observe? Have you road tested it personally to the same extent that you did with Sterculia Oil?
I bought some Alpha Lipoic Acid after reading this article (ordered some succin-ade too). Word or warning, it made my urine smell… not pleasant. Not quite asparagus unpleasant, but close.
Oh my…weeWee-ade
This was the ALA, not the Succin-ade. Started the S-ade on Saturday. Zero ill effects so far at 3x scoops a day. The ALA is still having the same effect.
Wanted to try this a year ago, but I couldn’t find any succinate so I got sodium propionate instead. I tried it for a little while and it seemed to help with weight loss a little, but it had an unpleasantly cheesy flavor.
As for succinate causing the runs, it’s probably just osmosis. Citric acid and citrate salts do the same thing. Magnesium citrate is even sold as a laxative.
Brad – Can I just double check the glucose FADH2/NADH Ratio with you!! So are we now saying that all Carbohydrates (Starch / Glucose / Fructose etc) are the same ratio but cause SCD1 values to differ??? Probably not but if you could give a quick explanation that would be amazing.
Glucose is normally 0.20 but under specific conditions when ROS is already high from saturated fat, an interesting rebalance of enzymes occur which result in glucose acting on FADH2 hence the “up to 0.5” claim.
To clarify, this was the subject of a recent post of Brad’s – https://fireinabottle.net/ampk-is-activated-by-high-cellular-energy-after-a-starchy-meal/
So I am not keen on the lemon limey monkfruit taste of succinate. Would it have a negative effect on the experiment if I just got plain succinic acid/succinate myself and cut it with baking soda to reduce the sour flavor? Would changing the PH like that render the succinic acid useless? Also, would this be good to try while fasting or should the succinate be taken with food?
I added some pure stevia powder to mine. It takes some of the bite away.
mixing some o.j in transformed it for me
Sodium succinate is much less sour, but also more expensive and less bang for the buck since is ~30% sodium. Be aware that most of the stuff out there is made from petroleum, if you care about that. I’m sourcing food grade disodium succinate that is made in a fermentation process, which is even more expensive and harder to find, but I feel like is worth it if we’re going to eat it!
Brad
Umm. My N=1 is not going well at all with the succinate. In the past week of using it, I have gone from 173 to 179. I have no idea why. My eating is the same, just with added succinate. I do take vitamin supplements. Is there maybe some interference that is going on from the vitamin supplements? I take Vitamin A, D, E, and K as well as a B complex. Thank you.
So there are a lot of factors at play: one is that ROS can stimulate AMPK and AMPK stimulates glycogen storage in muscle tissue. A six lb gain could easily be caused by glycogen uptake, but it is hard to tell. Are you measuring waistline as well?
Brad
Hey Brad – there was another commenter up there who asked the same question I have, which I understand you may or may not have the answer to, however, I am curious as to hear (read) your opinion on whether or not succinate would be beneficial while in a fasted state, and honestly, curious about whether Alpha Lipoic Acid would be as well. I was concerned with ALA that I might do something along the lines of possibly making me go hypoglycemic, but I did take a rather large dose the other day while fasted and it didn’t seem to have any negatives that I experienced.
I already got some of your succin-ade , I also have some stearic acid and your butter oil, and am curious, small amounts during fasted state… on any of this stuff, would theoretically increase fat burn, yeah?
These are interesting questions. I don’t necessarily have the answers to them. I’ve been thinking abut doing an n=1 of using ALA in the fasted state (first thing in the morning), then using the succin-ade an hour before meals. The idea is that the ALA will help reduce lipogenesis/activate sirtuins while I’m fasting, priming me to burn the succinate along with the meal to increase the effective F:N ratio of the meal. Is this the best approach? IDK, time will tell.
Just received my batch and getting ready to experiment! Does it have to be consumed before a meal or could it be used when fasting to work on existing unsaturated body fat?
It’s a little unclear at the moment the best strategy. I explained to another post that I am planning ALA while fasted and Succin-ade one hour before meals.
Hi Brad, So I am going all in as a guinea pig from today taking the following:
1 scoop of Succinade 1 hour before each of my 3 meals a day (3 scoops total)
2 squeezes of Sterculia Oil 15 mins before my first meal of the day.
2400mg of R-Alpha Lipoic Acid, fasted when I wake up each morning.
Vitamin D3 + K2 with Breakfast and Dinner
But I just wanted to know if I should also supplement with the following or stop them with the above in mind:
1g Berberine 15 mins before lunch and dinner (2g total)
15mg Astaxanthin after my dinner (15mg total)
Any pointers would be appreciated on all of the above. Thanks again Brad and just to say that I think your work on all of this is nothing short of genius!!!
Hi Lee! Your plan is nearly identical to what I’m going to try next month! Except I’ll probably only have two meals. Personally I would skip the berberine and astaxanthin, not because they’re not necessarily useful but because that’s too many variables to understand your result.
Thanks for the feedback Brad.
I am considering upping my succincade dose to 1.5 spoonful’s each meal for a total of 22.5g (3 x 1.5 scoops) Succinate a day. This is mainly based on my calories going up to about 3000 – 3500 a day now so I am guessing the higher succinate % of my calories would be helpful???
Perhaps, I’m interested in your results.
I’ve gone back to eating a lower fat, keto diet. Do I want to eat fat, particularly saturated fat, in the meals after the succinate intake?
Brad: “Also, gut bacteria generate a lot of succinate from fiber”
I wonder if that could explain why some gut bacteria are associated with weight loss, e.g. Akkermansia. Maybe they generate more succinate than other bacteria. I’ve seen some weight loss after eating a green banana every morning for a couple of months. Green bananas allegedly promote Akkermansia.
Brad, I have a question about Omega-3 supplementation from marine oils. I don’t take “fish oils”, I take Cod Liver Oil. I understand some would consider them to be the same, for the purposes of Omega-3 supplementation. I’ve seen some of your writings which indicate supplementing up to 3000mgs of Omega-3s (which I assume means a total EPA+DHA of 3000mgs total?) can be beneficial but I’ve also seen where they are potentially bad for a torpid metabolism due to them being highly unsaturated. I take a teaspoon (5ml, about 1000mgs combined EPA/DHA) each day, mostly to get vitamins A/D this time of year but also Omega-3s. I just ran out of the CLO and am wondering if it would be wise to just go without for a bit. Thoughts?
There is good evidence that long chain Omega-3 fats have the dual role of reducing lipogenic gene expression and increasing thermogenesis, at least in small amounts. I personally don’t take fish oil because I’m a wine drinker and fish oil plus ethanol is a bad combination for the liver.
My immediate question after reading the article, “What foods are high in succinate?”
Quick Anserwer: “Succinic acid is one of the natural acids found in foods such as broccoli, rhubarb, sugar beets, fresh meat extracts, various cheeses, and sauerkraut.”
Also young, non-mature fruits. <—-hello spring time exit from torpor?
And it looks like some wines are high as well.
Ok, so what "cheeses are high in succinate?" This one is a bit more research intensive but, right now its Emmentaller for the win. Some cheddars as well. I have giddy suspicions about
real parmesean.
Brad…I see a quiche here with broccoli, Emmentaller, some Firebrand bacon, … Thoughts?
Very interesting. Many bacterie excrete succinate as a waste product. We absorb it from gut bacteria. It makes sense that fermented foods would contain succinate. I haven’t had time to thoroughly research this.
Hi Brad,
Thank you so much for your blog, your experiments, research, and for sharing!
As a life-long weight-struggler, I was really excited to hear about this (well, I was mostly excited about the idea of eating croissants and bread again, not gonna lie). The science re: the saturated/unsaturated fats is shockingly missing from conversations about health (nothing like what we hear about sugar!), and so am very grateful to you for getting it out there to us all!
As a new member of the perimenopause gang, I’ve begun REALLY struggling with my weight. Weight which used to come off with dedicated exercise and low carb/high protein is going nowhere now. I’m trying this because the saturated fat concept makes sense to me, and so, I have mostly taken to eating plenty of dairy fat with starch (breads and potatoes mostly), and have gained (I think I expected it) approximately 7 lbs. I gained them all overnight, but have not gained since (will be starting my third week), so am assuming that it is water retention from the carbs. My energy levels are through the roof and as a long term runner, I am running faster and longer (and wanting to!) than I have in years. I feel that my temperature is running higher than it usually does, but to be honest have sadly not noticed inch (or scale) loss yet. Perhaps too much oleaic acid in my cells? Perhaps eating too much? Am thinking of a couple of things to change:
1. Return to intermittent fasting (one meal, late in the day). I think I am finally getting over the croissant/bread excitement and can start moderating how many croissants I actually need to eat as opposed to ‘want’ to eat (I discovered that I can eat a shockingly large number of them).
2. Adding some of your recommended supplements, but given the evolution of your theory, am a bit unsure as to what to add. Am thinking the succin-ade and the alpha lipoic acid? Should I also try some sterculia? Berberine?
Thanks again, and am looking forward to your next post. I’ve been binge reading them!
– Louise
Thanks for your comments! If I were to try one thing it would be ALA while fasted and Succin-ade before meals. Everyone’s biology is different, and yes, you probably have plentiful stored MUFA, which is a problem I have as well. The succinate should help with that, hopefully. Good luck!
I’ve failed with TCD (gain), stearic acid shenanigans, and sterculia. Just started this today. There’s a link to my spreadsheet elsewhere if anyone’s interested in keeping track of my progress.
Brad,
Thank you for providing this very saturated food for thought… one question on ALA use. Do you think Acetyl-L-Carnitine would have synergistic benefits with the succinate and ALA, ala (some pun intended) the work of Bruce Ames?
This question is actually on my radar, but I haven’t had time to research it yet!
https://journals.physiology.org/doi/full/10.1152/ajpendo.00094.2021?fbclid=IwAR1ET-izaDpcK5Uv8Y6f40f1VRKjmWtp7Gps4ezs5yeITwfcrxIeaEjGyQA
New study on carb over feeding and redox implications. Hoping this is on your radar, definitely a hyper lipid level of cryptic puzzle this one.
Wow this is neat. Brad you know I’m trying a McDougall-esque (non-vegan, though) high starch diet.
“Here, we show that acute COF is characterized by net transfer of electrons from mitochondria to cytoplasm. Circulating glutathione is oxidized in a manner that significantly cross-correlates with increasing insulin levels and precedes the decrease in cytoplasmic Eh. This effect is more pronounced in overweight individuals (OW).”
I’m reading this as: High glucose is firing up ROS. Just like yours & Peter’s posts read a while back. The missing piece in this article is the ROS-as-NAD+-regenerating-thermogenic-cycle part.
Instead they fold like a wet towel with the conclusion: “We conclude that acute COF contributes to metabolic dysfunction through insulin-dependent pathways that promote electron transfer to the cytoplasm and decrease antioxidant capacity. Characterization of redox during overfeeding is important for understanding the pathophysiology of obesity and type 2 diabetes.”
I guess part of why they missed the NNT point here is they’re using a hyperinsulinemic clamp – they are intentionally infusing insulin which would trash the thermogenic process and prevent one from seeing the insulin-like effect of AMPK (GLUT4 recruitment) under the hyperglucose condition.
It’s not all great, though. The obese have more oxidized glutathione, suggesting that they aren’t recycling NAD+ as quickly…
Thanks for the link, that’s actually a fascinating paper! I’ll be writing about it…
Hi Brad,
If succinate has an F/N ratio of 1.0, what are the F/N Ratios of other Kreb’s cycle intermediate like Fumarate, Malate, Citrate?
I know citrate is a common food additive.
Good question!
Citrate: 0.33
a-ketoglutarate: 0.5
Malate/Fumarate: 0
I doubt there is enough added citrate to effect an overall meal.
Started this morning. Succinade and alpha lipoic acid. https://docs.google.com/spreadsheets/d/1H9myetpWoyw3acL4aKjUk4lc9xxxf5hF80poyqZjj1A/edit?usp=sharing
Wow, thanks for sharing!
Add your morning blood pressure, it increased for me
I got 2 bags and am doing 2 scoops a day plus ALA each morning. Not looking to lose massive weight, but lean out last few pounds. In the past, I have done Eca stacks minus the aspirin with some success. What are your thoughts on running both Eca and Succinade concurrently? Am I asking for trouble?
It might be a lot all at once but I’m not the boss of you.
Adding my approach to and experience with taking Succin-ade. Short version: am taking much less than suggested while having great results. Am a 62 year old woman in good overall health but had a prediabetic blood test a few years ago so started eating low carb. I had clear blood sugar issues since my 20s (hypoglycemia, eating 5x a day, excessive thirst, fatigue) just wasn’t bad enough to meet the “official” b.s. criteria until my 50s. Low carb improved my numbers but I still had poor muscle tone, low energy most of the time, and bouts of extreme fatigue.
After hearing about possible headaches, high blood pressure, and/or laxative effects, I wanted to start cautiously with the Succin-ade. Used 1/2 teaspoon in 8 oz of water with 100 mg of alpha-lipoic acid in early morning. Felt full as if I had already eaten (normally I eat breakfast without fail.) Had 2 meals that day instead of 3. Have tried even less Succin-ade since, have been taking 1/4 teaspoon per day. Now have much more energy that lasts all day, can skip breakfast or dinner on many days, appetite is lower than ever. I usually eat a carnivore 1st meal and a keto 2nd meal, usually. No weight loss so far, have not focused on that but would like to trade in 15 lbs of fat for 5 pounds of muscle.
I may try increasing the Succin-ade but so far so good. Really good, actually. Energy = life.