This is another article in my series on reductive stress as a unifying model for obesity. The intro article is here!
Food Is Reduced
In the food that we consume, the Hydrogens (H) and Carbons (C) are in a reduced state – they are in control of their own electrons. We release them as CO2 and H20: in these molecules the Cs and Hs are fully oxidized. Oxygen is in control of their electrons. During metabolism, the electrons flow from C and H to O. The electrons are typically carried by the oxidized NAD+, which becomes NADH in its reduced (electron carrying) form.
When the metabolic rate in a cell proceeds beyond ATP demands, NADH can build up at the expense of NAD+. Too many things are reduced and not enough are oxidized. This is reductive stress.
Reductive stress looks like energy overload to the cell. There is tons of NADH around with nowhere for the electrons to flow to. Remember that the source of NADH was the food that you are burning. Too much NADH looks like too much food from the cellular perspective.
There are (at least) five things the cell can do with the extra electrons. Remember that enzymes such as NNT can convert NADH to NADPH, so the two pools are somewhat interchangeable.
- NADH can donate electrons to the Electron Transport Chain (ETC) to create more ATP as needed.
- Electrons can spew from the ETC to become the Reactive Oxygen Specie superoxide.
- NADPH oxidase can use electrons from NADPH to create superoxide.
- The cell can import pyruvate, use the electron from NADH to convert the pyruvate to lactate, then export the lactate for another cell to use.1
- The cell can use the reducing power of NADPH to convert glucose to fat using the process of De Novo Lipogenesis (DNL)
Fat From Glucose
The Cs and Hs in fat are in a much more reduced state than the ones in glucose. Glucose – C6H12O6 – has one oxygen for each carbon. Palmitic acid – the 16-carbon saturated fat – is C16H32O2: one one Oxygen for each 8 Carbons. This fact has some real world consequences. Oxygen being the electron strongholder that it is, has a tiny negative charge when bonded to Hydrogen and the Hydrogen has a tiny positive charge. This is what makes glucose soluble in water – an ideal circulating fuel for plants and animals. This is also the reason why fat has double the calories per gram than glucose – glucose is already halfway oxidized. The higher energy density of fat makes it an ideal storage fuel for animals who have to carry their energy cache on their back.
The point is that if you want to convert glucose to fat, you have some work to do. You need reducing power and that is provided by NADPH both via glucose sent through the pentose phosphate pathway in the cytosol or via an enzyme called the malic enzyme which is used to export reductive stress from the mitochondria to the cytosol. So you can donate electrons from one molecule of glucose to another molecule of glucose to turn it into fat if the cell is in reductive stress: NADPH is in high supply.
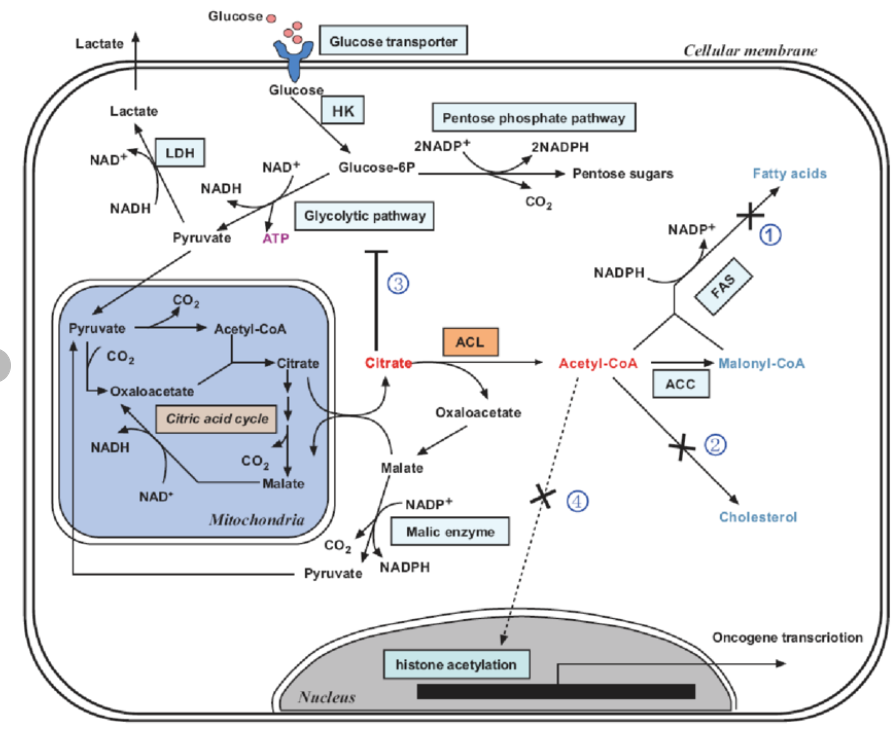
In the last article I talked about mice who had NOX4 (NADPH Oxidase 4) removed from their hearts, which put the heart cells into reductive stress. NOX4 literally just takes the electrons from NADPH and turns them into sueroxide, eliminating the reductive stress. If you were to remove NOX4 from the whole body of a mouse, you might predict whole body reductive stress to cause increased DNL. In fact, if you remove NOX4 from a mouse, it will become fat on the starch based chow diet that keeps most mice lean in the lab. 3
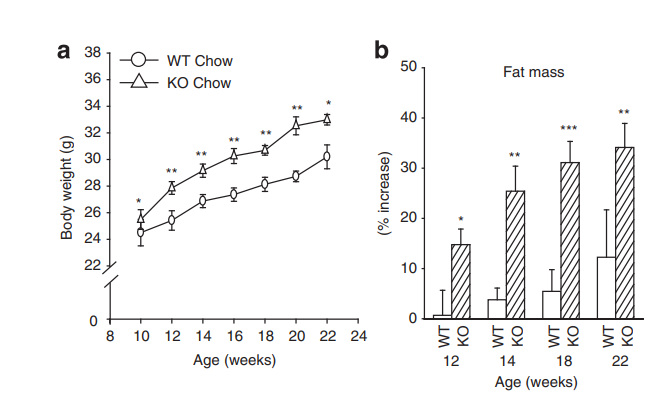
Reductive Stress Causes Fat from Carbohydrates
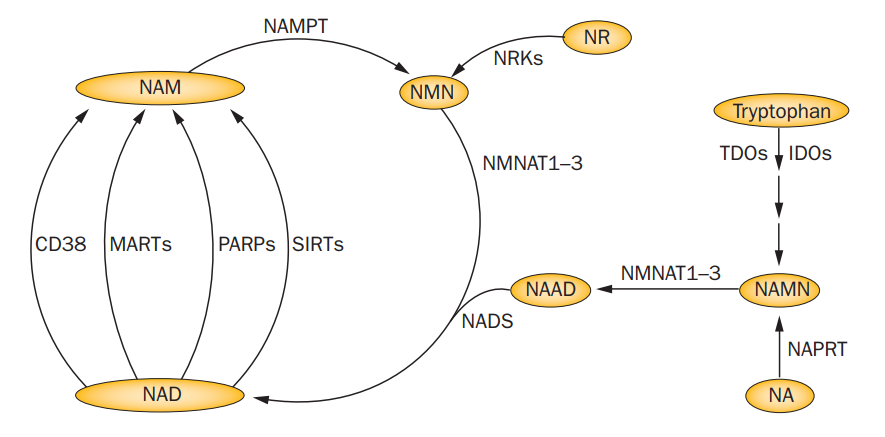
The relative levels of NAD+ are affected by a number of enzymes, the most important of which are CD38 and NAMPT. CD38 uses NAD+ as a co-factor, converting it to (a variety of things plus) Nicotinimide (NAM) and drawing down the NAD+ pool. NAMPT is the rate limiting enzyme that recycles NAM back into NAD+.
Reductive stress in the NAD+/NADH redox couple, can therefore be created either by increasing CD38 activity or by decreasing NAMPT activity.4
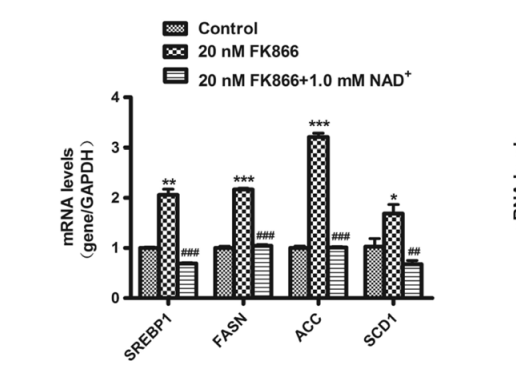
This paper5 has a really nice demonstration using an NAMPT inhibitor called FK866 to show that in fact lipogenic gene expression is controlled by reductive stress itself. The model system was mouse liver cells grown in culture. The genes that were looked at were the lipogenic transcription factor SREBP-1c plus the enzymes Fatty Acid Synthase, ACC and our old friend SCD1, all of which are highly involved in DNL. The authors showed that inducing reductive stress by blocking NAMPT activity increased the expression of the lipogenic genes and that adding NAD+ reversed the increase.
The expression of lipogenic genes is controlled by the level of reductive stress in the NAD+/NADH redox couple!
Check back soon, I’m working on a post about CD38, then we’ll start to look at the role of vegetable oils and what we can learn from obesogens.
- 1.Rabinowitz JD, Enerbäck S. Lactate: the ugly duckling of energy metabolism. Nat Metab. Published online July 2020:566-571. doi:10.1038/s42255-020-0243-4
- 2.Zu XY, Zhang QH, Liu JH, et al. ATP Citrate Lyase Inhibitors as Novel Cancer Therapeutic Agents. PRA. Published online March 1, 2012:154-167. doi:10.2174/157489212799972954
- 3.Li Y, Mouche S, Sajic T, et al. Deficiency in the NADPH oxidase 4 predisposes towards diet-induced obesity. Int J Obes. Published online March 20, 2012:1503-1513. doi:10.1038/ijo.2011.279
- 4.Garten A, Schuster S, Penke M, Gorski T, de Giorgis T, Kiess W. Physiological and pathophysiological roles of NAMPT and NAD metabolism. Nat Rev Endocrinol. Published online July 28, 2015:535-546. doi:10.1038/nrendo.2015.117
- 5.Wang LF, Wang XN, Huang CC, et al. Inhibition of NAMPT aggravates high fat diet-induced hepatic steatosis in mice through regulating Sirt1/AMPKα/SREBP1 signaling pathway. Lipids Health Dis. Published online April 27, 2017. doi:10.1186/s12944-017-0464-z

Hi Brad;
How does cyproheptadine make you gain weight?
Does cyproheptadine also cause stress reduction?
Interesting question. I don’t know that much about it, but it affects tryptophan metabolism and that’s the first place I’d look. The tryptophan metabolite kynurenine is an Aryl Hydrocarbon Receptor (AhR) agonist. AhR agonists definitely cause reductive stress, which is the topic of an upcoming post.