In this series, I have argued that consuming seed oils causes reductive stress in the mitochondria as defined by high levels of acetyl-CoA and NADH. You could also call this “too much fuel” in the mitochondira, AKA a flooded engine. If I am correct, it should be easy to demonstrate in the short term that consuming a large quantity of seed oils causes acute reductive stress.
Pyruvate Dehydrogense
A hallmark of human obesity is the lack of ability to efficiently burn glucose1. The enzyme that is the gating factor for how quickly glucose can be burned is pyruvate dehydrogenase (PDH). PDH converts pyruvate – the end product of glycolysis – to acetyl-CoA to be burned in the TCA cycle.
Way back in 1975, it was already known that PDH activity was inhibited by high levels of acetyl-CoA and NADH2 – reductive stress. If you are in reductive stress, PDH is inhibited and you become inefficient at burning glucose: like a fat person. Additionally, Cody Smith recently showed that overexpressing PPAR alpha – an enzyme that is activated by the linoleic acid and a-linolenic acid in seed oils – floods the mitochondria leading to whole body reductive stress and glucose intolerance.3 Things that overstimulate PPAR alpha cause reductive stress and glucose intolerance.
Let’s consider for a moment what should happen when a healthy person eats a mixed meal of fat and carbohydrate. Insulin is released, which cuts off lipolysis (fat being released by fat cells to feed the body) and increases glucose uptake and oxidation rates. After a mixed meal, you are supposed to burn the glucose first to prevent high post-prandial blood glucose. Lean people are better at this than fat people.4 The lean children in this study had a post-prandial RER of 0.92 indicating they were mostly burning carbs after a mixed meal. The obese children had an RER of 0.87, meaning they were burning way less carbs in response to a mixed meal.
In 1988 – a mere 13 years after it was reported that NADH and acetyl-CoA block PDH activity – this study was published showing that eating a mixed meal with PUFA, as opposed to with saturated fat, caused lower rates of carbohydrate oxidation after the meal. Just like in the fat kids!!
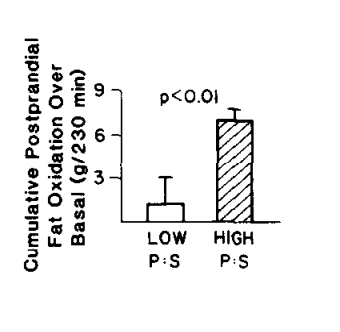
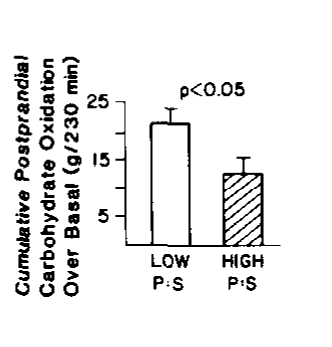
Promoters of seed oil point to studies like this and say, “See, PUFA increase fat burning rates! And total calories burned!”
Which is true, but you could also say, “See, PUFA increase fat burning rates! Just like fat people! And total calories burned! Just like fat people!”
It looks to me like PUFA, by stimulating PPAR alpha, are driving an inappropriate amount of fuel into the mitochondria and creating reductive stress compared to saturated fat. The buildup of acetyl-CoA and NADH are inhibiting PDH and therefore the lean people given PUFA are behaving like fat people, who are also in reductive stress.
Sunflower Oil Inhibits PDH
An experiment tested the effect of fat loading on exercise-stimulated PDH activity5. The subjects are all young men who are very physically active. We are not told their BMI, but the suggestion is that they are lean. Each subject repeated the trial, once by fat loading with cream and once by fat loading with sunflower oil. Muscle biopsies were taken before fat loading, after fat loading and after a minute of exercise.
PDH activity was unchanged during fat loading but after exercise PDH activity surged in the group given heavy cream. This group appropriately increased blood glucose burning in response to exercise. The sunflower oil group failed to increase glucose burning in response to exercise.
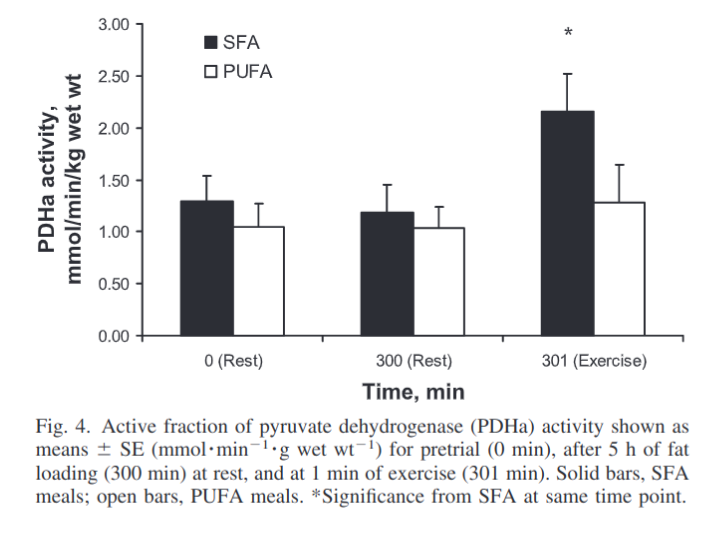
Just like old, obese, insulin-resistant humans.6
Conclusion
Obese humans are characterized by a lack of metabolic flexibility. They have low pyruvate dehydrogenase activity and therefore fail to switch over to efficiently burning blood glucose after exercise or a mixed meal. Lean adults can mimic the lack of metabolic flexibility shown by obese adults simply by consuming enough PUFA, which forces them into reductive stress.
Check back in soon, article VI of the “Seed oils cause reductive stress” series will be the last one. It is about PUFA acting as an obesogen. I’m excited for it and then we will get to the hard work of using all of this knowledge to fix reductive stress in ourselves.
- 1.Istfan N, Hasson B, Apovian C, et al. Acute carbohydrate overfeeding: a redox model of insulin action and its impact on metabolic dysfunction in humans. American Journal of Physiology-Endocrinology and Metabolism. Published online November 1, 2021:E636-E651. doi:10.1152/ajpendo.00094.2021
- 2.Pettit FH, Pelley JW, Reed LJ. Regulation of pyruvate dehydrogenase kinase and phosphatase by acetyl-CoA/CoA and NADH/NAD ratios. Biochemical and Biophysical Research Communications. Published online July 1975:575-582. doi:10.1016/s0006-291x(75)80185-9
- 3.Smith CD, Lin CT, McMillin SL, et al. Genetically increasing flux through β-oxidation in skeletal muscle increases mitochondrial reductive stress and glucose intolerance. American Journal of Physiology-Endocrinology and Metabolism. Published online May 1, 2021:E938-E950. doi:10.1152/ajpendo.00010.2021
- 4.Aucouturier J, Isacco L, Thivel D, et al. Effect of time interval between food intake and exercise on substrate oxidation during exercise in obese and lean children. Clinical Nutrition. Published online December 2011:780-785. doi:10.1016/j.clnu.2011.03.011
- 5.Bradley NS, Heigenhauser GJF, Roy BD, et al. The acute effects of differential dietary fatty acids on human skeletal muscle pyruvate dehydrogenase activity. Journal of Applied Physiology. Published online January 2008:1-9. doi:10.1152/japplphysiol.00636.2007
- 6.Prior SJ, Ryan AS, Stevenson TG, Goldberg AP. Metabolic inflexibility during submaximal aerobic exercise is associated with glucose intolerance in obese older adults. Obesity. Published online October 16, 2013:451-457. doi:10.1002/oby.20609

I thought that it was PPAR-gamma that got activated by PUFA, as mentioned in https://fireinabottle.net/vitamin-d3-and-k2-activate-ampk/?
Both PPARs, in fact I think all three PPARs are activated by PUFA. The ramifications of this are complicated. Probably sometimes good/sometimes bad.
I was confused a bit by the “alpha bad” in this case as well.
This is probably a U-shaped curve. In a healthy animal, overstimulating PPAR alpha will send too much fat into the mitochondria. Especially if this is PUFA, reductive stress will ensue. You can send mice into torpor simply by giving them sunflower oil and a PPAR alpha activator.
Once you’re fully into torpor, PPAR alpha levels drop to well below baseline. This is presumably to preserve energy for hibernation. Once you’re fully torpid it makes sense to do something to get PPAR alpha back to baseline levels.
Brad
Okay. So this makes a lot of sense.
My husband started taking Astaxanthin at the end of April. Since then he has steadily gained weight (178-186 Lbs) with no other changes to his routine *except* that I have been fasting a lot myself and not feeding him.
So in combination with Astaxanthin his diet has gone from a tremendous amount of saturated fat (I take TCD fully to heart LOL) to a “diet” of mostly his own body fat during the day. He’s about 30% PUFA and I want to say 40% MUFA on his omega quant. So big difference in the fuel his mitochondria are getting in the two scenarios.
I’ve pulled the Astaxanthin for now and he can revisit it when I’m feeding him properly again.
So glad you cleared this up.
Hi Brad,
Lee again. Thanks as always for your help with everything, I think your work on all of this is nothing but outstanding!!
You mentioned about focusing on NAD+. So Would you suggest NMN or NR to help increase my metabolic rate even further ???
I already take Sterculia Oil with my lunch / Succinade before every meal / R-ALA in the morning and before bed + Apigenin before bed.
My gut feeling is to try NR over NMN and take out the Apigenin but what would you suggest??? What dose would you recommend for the NR?? And would you agree to taking out the Apigenin or do you think combining both could be even more potent??
Lastly, I thought I would leave some feedback on the products I have so it might help others:
The ALA works very well!! I have tried both the R- ALA and the cheaper S form. The R is outstanding with immediate effect and little to no side effects. The S form I found made me feel like my energy was being sapped away from me / very lethargic.
Apigenin I am unsure about!! I think it is helping but despite taking a BIG dose each night before bed, I wouldn’t say it’s as obvious as other supplements for its effect.
Succinade is amazing!!! Expensive as hell when taking a big dose each day like I do but so obvious in its effects. I also feel great when I am on this stuff and that my strength training is much easier.
Sterculia Oil – Hard to say if it’s the main reason for it but the body fat I still have feels VERY firm in comparison to 6 months ago.
All in all I feel with Brads help I am winning a war against being fat that I had been losing for many years!!! I eat 4000 calories a day and I am still losing body fat slowly but surely every week!! Listen to this guy, he is definitely onto something here.
Hey Lee!
Thanks for the feedback! In most of the papers I’ve seen, NR works a little better than NMN. I’m currently taking 300mg of NR night and evening (600mg total). It’s expensive but worth it, I think.
As to apigenin, it’s hard to say. It probably also depends on your personal situation. If you have leaky gut issues, blocking CD38 might be more important than if you have upregulated AhR, which drains NAD+ by upregulating PARP instead of CD38. OTOH, apigenin blocks the AhR as well, so it sounds very good on paper! My hesitation is – and I’ve somehow lost the link to the paper – there was a study that flavones (I think naringenin) interfere with NAD metabolism. Which makes sense since they block CD38’s NAD binding site (presumably).
Rodent studies with apigenin look very good, however. I’d say try two weeks with and two weeks without?
Brad