Overview
- Both PPAR alpha and PPAR gamma reduce diabetes symptoms
- PPAR gamma causes weight gain in humans
- PPAR alpha causes weight loss in humans
- Stimulating PPAR alpha reverses hi LA induced obesity in mice; Stimulating PPAR gamma does not
- Starch eating mice make lots of PPAR alpha and less PPAR gamma
- Mice whose diets have over 6% of calories from linoleic acid (LA) have a large decrease in PPAR alpha and a large increase in PPAR gamma
- PPAR gamma upregulates the “lipogenic” (fat making) genes SCD1, Elovl3 and FAS.
- The end products of SCD1, Elovl3 and FAS upregulate PPAR gamma. It’s a vicious cycle.
- Some options are presented.
Introduction
I introduced PPAR alpha in Dietary Monounsaturated Fat as an altogether good actor, doing things like increasing the rate of fat burning. The PPAR genes are indeed master metabolic regulators. In this post I’m going to introduce PPAR alpha’s troubled sibling, PPAR gamma.
The PPARs are transcription factors, which is to say that when they are produced, they turn other genes on or off. In fact, there is a huge amount of overlap in the genes turned on by both PPAR alpha and gamma, including genes involved in fat transport, fat oxidation, fat creation (lipogenesis), adipocyte differentiation, adaptive thermogenesis (mitochondrial uncoupling) and others. SInce they have such a broadly overlapping set of target genes and those genes are involved in so many processes it can be hard to tease out exactly what makes them distinct. But on the aggregate we know that they behave differently.
For instance, increasing PPAR alpha seems to aid in weight LOSS, whereas stimulating PPAR gamma most definitely causes weight GAIN. We know that because a PPAR gamma agonist – which means something that turns PPAR gamma ON – called rosiglitazone, is used to treat diabetic symptoms. One study stated quite candidly that “Weight gain was the main adverse effect”1 of rosiglitazone. So increasing PPAR gamma causes weight gain in humans. On the contrary, the Chinese herb berberine is a powerful PPAR alpha agonist2 and it causes weight loss in humans.3
The Standard American Diet Increases PPAR gamma and Decreases PPAR Alpha
In The Body Fat of Starch Eaters, I point out that healthy starch eating cultures have very saturated body fat, high metabolic rates, consume a lot of calories and stay lean. But what happens when starch eaters eat too much linoleic acid?
In this study,4 (a strain of prone to obesity) mice were put on a starch based diet containing ~5% linoleic acid (LA), which we’ve seen is a safe amount if you are metabolically healthy, or they were put on a “hi-fat” diet where ~10% of calories were from linoleic acid (assuming the lard was 15% LA), an amount that we know will lead to dys-regulated metabolism.
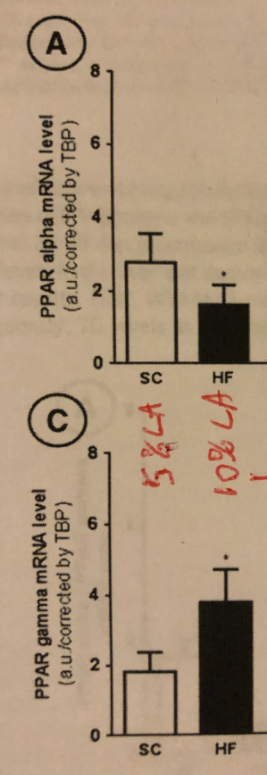
The mice eating a low LA diet were producing 50% more PPAR alpha than they were PPAR gamma. The dys-regulated mice were producing 2.5 TIMES as much PPAR gamma as PPAR alpha!! Going over 5-6% of LA in your diet seems to be a switch that turns on PPAR gamma and turns off PPAR alpha. This is likely useful to a hibernating bear eating acorns to fatten up for winter but likely less useful for you. I suspect Native Americans were aware of the metabolic changes associated with LA overconsumption and that is why they replaced acorn oil with low-PUFA, spring-collected bear grease.
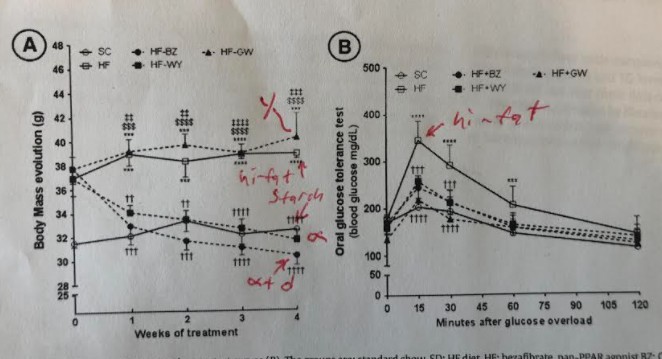
Other mice were put on the hi LA diet and given pharmaceuticals that would upregulate either their PPAR gamma, their PPAR alpha or both PPAR alpha and a third member of the family called PPAR delta.
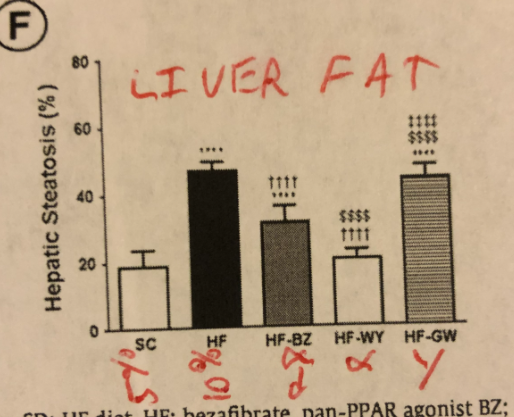
The mice on the hi-fat, hi-LA diets became obese as expected. The PPAR alpha enhanced mice, who were obese at the start of the drug intervention, rapidly lost weight and weighed less than the starch fed mice at the end and had the same amount of liver fat as control mice. The PPAR gamma enhanced mice were if anything a little fatter than the high fat diet fed mice at the end and had just as much liver fat!
Interestingly, the mice given the PPAR agonists – for either gamma OR alpha – had better glucose tolerance than mice on the high fat diet alone. The difference is in how they got there.
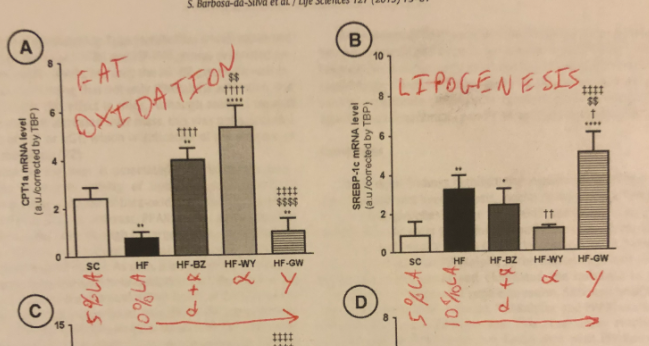
PPAR gamma helps reduce excess circulating energy by helping to STORE fat. PPAR alpha helps reduce excess circulating energy by helping to BURN fat.
A Vicious Cycle
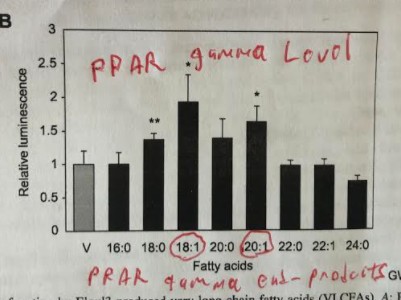
One of the things that PPAR gamma does is increase lipogenesis (making fat). It does this by increasing the expression of several genes including our old friend SCD1 in addition to fatty acid synthase (FAS) and elongase 3 (Elovl3). FAS uses available acetyl groups (from starch, fructose or ethanol) to build into palmitic acid – a 16 carbon saturated fat. Elovl3 extends palmitic acid into the 18 carbon length stearic acid (18:0) or even the 20 carbon length arichidic acid (20:0). SCD1 desaturates these into the monounsaturated oleic acid (18:1) and the monounsaturated eicosenoic acid (20:1).
Here’s where it gets interesting and tragic. The end products of upregulated PPAR gamma – oleic acid and eicosenoic acid – upregulate PPAR gamma.5 This is a positive feedback loop. Once you start making a lot of PPAR gamma you will continue to make it.
How Can One Tell If One Makes a Lot Of PPAR gamma?
Since the production of PPAR gamma and SCD1 are tightly correlated, the Desaturase Index (DI) is a good indicator of upregulated PPAR gamma. I introduced the DI in The SCD1 Theory of Obesity Part 2. In brief, it is the amount of oleic acid in a tissue divided by the amount of stearic acid. Since SCD1 – which is upregulated by PPAR gamma and whose end products upregulate PPAR gamma – converts stearic acid to oleic acid the ratio of the two can tell you if your PPAR gamma is dys-regulated.
If you take the OmegaQuant complete test, you will be able to get your oleic and stearic acid numbers. Ideally you would have 1.6 times as much oleic as stearic or less.
I’ll have a lot more info about the DI in the next few weeks, so check back in.
How can I Stimulate PPAR alpha While Reducing PPAR gamma?
Perhaps not coincidentally, a lot of the things I’ve recommended on the blog that reduce SCD1 expression, thus hopefully breaking the cycle of more SCD1 leading to more PPAR gamma leading to more SCD1 – ALSO increase PPAR alpha. The two PPARs seem to be inversely regulated in many cases. As I’ve already pointed out, berberine is a powerful PPAR alpha agonist that also downregulates SCD1. Fish oil is another.6 AOE is a third. CLA is another.7
It is worth noting that David, who just submitted a MOST impressive Croissant Diet testimonial, used fish oil supplements during his weight loss, stimulating PPAR alpha and suppressing SCD1.
I’ll also be presenting multiple lines of evidence that sterculia oil was able to downregulate PPAR gamma in myself in the coming weeks. Getting the sterculia oil imported and bottled has been an epic challenge but we should be shipping within the next two weeks.
- 1.Ratziu V, Giral P, Jacqueminet S, et al. Rosiglitazone for Nonalcoholic Steatohepatitis: One-Year Results of the Randomized Placebo-Controlled Fatty Liver Improvement With Rosiglitazone Therapy (FLIRT) Trial. Gastroenterology. Published online July 2008:100-110. doi:10.1053/j.gastro.2008.03.078
- 2.Zhou Q. Berberine is a potent agonist of peroxisome proliferator activated receptor alpha. Front Biosci. Published online 2016:1052-1060. doi:10.2741/4440
- 3.Hu Y, Ehli EA, Kittelsrud J, et al. Lipid-lowering effect of berberine in human subjects and rats. Phytomedicine. Published online July 2012:861-867. doi:10.1016/j.phymed.2012.05.009
- 4.Barbosa-da-Silva S, Souza-Mello V, Magliano DC, Marinho T de S, Aguila MB, Mandarim-de-Lacerda CA. Singular effects of PPAR agonists on nonalcoholic fatty liver disease of diet-induced obese mice. Life Sciences. Published online April 2015:73-81. doi:10.1016/j.lfs.2015.02.003
- 5.Kobayashi T, Fujimori K. Very long-chain-fatty acids enhance adipogenesis through coregulation of Elovl3 and PPARγ in 3T3-L1 cells. American Journal of Physiology-Endocrinology and Metabolism. Published online June 15, 2012:E1461-E1471. doi:10.1152/ajpendo.00623.2011
- 6.Bargut TCL, Mandarim-de-Lacerda CA, Aguila MB. A high-fish-oil diet prevents adiposity and modulates white adipose tissue inflammation pathways in mice. The Journal of Nutritional Biochemistry. Published online September 2015:960-969. doi:10.1016/j.jnutbio.2015.04.002
- 7.Moya-Camarena S, Vanden H, Blanchard S, Leesnitzer L, Belury M. Conjugated linoleic acid is a potent naturally occurring ligand and activator of PPARalpha. J Lipid Res. 1999;40(8):1426-1433. https://www.ncbi.nlm.nih.gov/pubmed/10428978

Nice, I enjoy reading you, Thank you
I live in Latin America (Panama City), it is possible to import some of your products here?
I ship to Latin America. The shipping rates are $12 US for up to a pound, $20 for two lbs or $30 for up to four lbs.
Brad
Brad,
Great article! I was just reading this here: https://pubmed.ncbi.nlm.nih.gov/10428978/
, It seems that Conjugated linoleic acid (CLA) is a potent up regulator for PPAR alpha as well.
I’m considering adding in some L-carnitine, and Rauwolscine an alpha-2 adrenoceptor antagonist
https://pubmed.ncbi.nlm.nih.gov/12378044/
https://www.sciencedirect.com/science/article/pii/B9780128012383988615
David
Good find! I’m going to add it to the article.
Brad, great article as usual.
I wanted to see what your thoughts were on fully-hydrogenated oils.
If we were to fully hydrogenate ghee, you’d take the 28% of fatty acids that are either 18:1, 18:2, or 18:3 and turn them into stearic acid, changing the total stearic acid content from 11% to 39%.
This would technically be the ideal cooking fat, no?
I know you can buy fully hydrogenated coconut oil, but that’ll still have a ton of short-chain fatty acids which we’re unsure about.
I think there’s a hydrogenated palm oil product that’s popular in India, but it comes in both partially- and fully-hydrogenated versions.
But then you’d ALSO have the palmitic acid content of ~28%. I’m pretty sure that would be inedibly waxy.
Even if you were to use it exclusively for frying?
Perhaps… Hmm, I think all you’d need is a nickel catalyst and a tank of pure Hydrogen gas…
Brad
Brad, I got my OmegaQuant results on Jan 7.
How do you recommend spacing out these tests to learn if you’re on the right path?
It’s a good question. I’ve been testing every 2-6 weeks, depending on what I’m doing dietarily/with supplements. You can see a change in two weeks for sure, but obviously that’s expensive. Perhaps every 4-6 weeks or even quarterly is a reasonable compromise?
Brad
I’m losing weight on meat, some veggies, a little rice. Salads with olive oil an balsamic. Occasional full fat cottage cheese and Bulgarian yogurt. Fresh pears and low sugar berries. But these days eating croissant filled with eggs and cheese which is very filling. Wash down with cup pu’er tea and 500-1000mg berberine/day.
Only supplements D3, K7, DHA
This is working but not yet able to say sustained weight loss yet. Two meals a day. Not hungry. Stopped afternoon snacking. Occssional carb crave near bedtime but can resist. Delaying follow up OQ until I sop or slow down weight loss
Hey Hap!
Excellent news, keep us updated!
Brad
Hi Brad,
When to take the supps? With a meal or fasted?
I’m taking:
Fish oil 3g
Berberine 2.4g
Jiagulan Ectract 1.6g
Hesperidin 0.5g
CLA 3g
Wondering if my doses are accurate too? Been taking 1 week so far. Don’t notice any difference yet.
I’m 15% body fat hoping I can get more lean.
Thanks for the input.
Max
I would suggest taking them with meals. I’ll be curious if you get a response from those. Are you tracking body temp and/or waistline?
Brad
I am tracking body temp but I’m guessing I won’t see any noticeable changes until months of it. So far I’ve noticed less of a need to go to the bathroom in the middle of the night. I like to drink water before I go to bed so atleast I’m getting some benefit so far.
My waistline is already pretty narrow but I want to see if some annoying stubborn fat deposits would disappear.
Max