Introduction
It’s time for the season finale! Next week I will pivot to discussing my personal results of using sterculia oil to inhibit SCD1. In this post I will summarize my posts on the topic to date before bringing the thread to a near term conclusion using an example that in retrospect will be so obvious that you’ll kick yourself for not having guessed it: the Syrian hamster, a food-storing hibernator.
The story of the Syrian hamster and the way it juggles its metabolism to hibernate is a beautiful narrative example of the topics covered in this thread. The teaser version is that to get into a state of deep torpor – low metabolic rate, lowered body temperature – it elevates SCD1 and PPAR gamma. Does this sound like anyone you know? Once the hamster reaches the state of deep torpor it becomes very good at maintaining its fat mass with very little food.
Deep torpor is a state that many mammals have the ability to go into: dormice, hamsters, bears, bats, woodchucks, chipmunks, lemurs, hedgehogs, skunks, and on and on. This suggests that the basic metabolic tools to get into deep torpor were in place before mammals branched off of the evolutionary tree. If you’re reading this you’re likely to be a mammal. Are you in deep torpor?
Before we jump into it, let’s make sure everyone is up to date.

Summary Of The SCD1 Theory Of Obesity To Date
So far, in this season of Fire In A Bottle:
- The SCD1 Theory Of Obesity Part One: Leptin is a hormone released by fat cells. As you get fatter you make more of it. The job of leptin is to increase the rate of fat burning which drives mitochondrial Reactive Oxygen Species (ROS) production. Mitochondrial ROS suppresses production of SCD1, whose job is to turn saturated fats into monounsaturated fats. When your fat becomes too unsaturated, you fail to generate ROS production. If you generate enough ROS your fat cells will go through a process called browning, which leads to thermogenesis – a process by which you burn calories off as heat. If you make too much SCD1, your fat won’t be saturated enough to do this and you will stop responding properly to leptin. It has been shown very clearly that mice who lack leptin become very fat but that if they ALSO lack SCD1 they are nearly as lean as mice who have leptin. Leptins main job is to suppress SCD1. Things that lack SCD1 fail to store fat and things that make lots of SCD1 get fat, including Native Americans living an ancestral lifestyle but eating nothing but maple sugar.
- The SCD1 Theory of Obesity Part 2: A LOT of ROS inhibits SCD1 but a LITTLE ROS increases it. The consumption of too much linoleic acid (LA), a common omega 6 polyunsaturated fat (PUFA), leads to a LITTLE mitochondrial ROS production. This leads to SCD1 being upregulated which leads to your fat being unsaturated which leads to a little ROS which leads to more SCD1. A positive feedback loop of unsaturation and failure to do thermogenesis. This is the post-obese metabolism. Many of us are in this boat. You can diagnose this using a blood test that measures the amount of oleic and stearic acid in your red blood cell phospholipids. Just divide oleic by stearic to get your Desaturase Index (DI). A lower DI is better. Things that suppress SCD1 include fish oil, CLA, metformin and berberine. Sterculia oil, from a tropical tree seed, blocks the enzymatic activity of SCD1.
- Membrane Phospholipid Composition of Different Populations shows that an ideal DI might be as low as 1.1.
- Dietary Monounsaturated Fat, The SCD1 Theory of Obesity, Part 3: There is a mechanism that the metabolism uses to cope with dietary monounsaturated fat (MUFA), which cannot be stored as is lest it trigger the post-obese metabolism. Oleic acid is turned into a signalling molecule called OEA in the intestine, which activates a transcription factor in fat cells called PPAR alpha AND it downregulates SCD1. PPAR alpha upregulates all areas of fat metabolism: lipogenesis, fat transport and fat oxidation. In this way saturated fat is produced to “up-saturate” the MUFA before it can be stored. The first pantry items in Africa 170,000 years ago were stored marrow bones and potato like tubers. Bone marrow is mostly MUFA. When eaten together, the starch from the tubers would have provided substrate for saturated fat to be produced to be blended in with the marrow fat.
- Pu’erh Tea is a potent Suppressor of SCD1: pretty much what it sounds like.
- This is Your Body Temperature on Vegetable Oil introduces a Bolivian population who had a well documented 1 degree F drop in body temperature since 2004. In 2004 they ate what they grew – manioc, plantain and rice: mostly starch – and had the classic body temperature of 98.6. A store was introduced after that giving them access to vegetable oil. A mouse model shows how adding PUFA to a starch based diet can cause a dramatic body temperature drop by lowering the amount of uncoupling protein (UCP1) that is produced which allows for mitochondrial uncoupling and adaptive thermogenesis – the ability to burn off calories as heat. The body temperatures of peoples of the industrialized world have dropped since the industrial revolution while vegetable oil consumption has risen.
- Speed Your Metabolism With Oxidative Stress! Slow It With Antioxidants! Mice lacking Nrf2 – the transcription factor responsible for antioxidant response – have frank oxidative stress, a high metabolic rate and resistance to obesity. Without the ability to reduce hydrogen peroxide, they are forced to produce uncoupling protein. This makes their metabolism “inefficient” – they burn off their calories as heat rather than storing them as fat. It’s funny. If you give them antioxidants, they no longer are forced to uncouple and they get fat.
- Native Americans Removed Acorn Oil and Replaced It With Bear Fat: There is documentary evidence that Native Americans removed the (relatively high PUFA) oil from acorns – a dietary staple. Yet they had no problem combining animal fats with starch from acorns, corn, and native potatoes and wild rye.
- The Body Fat Of Starch Eaters is Highly Saturated; Linoleic acid Dysregulates SCD1: Starch eating cultures have very saturated bodyfat. This explains this quote from the authors of the original China Health Study, “In the China Study, the least active Chinese consumed 30% more calories than their American counterparts yet their body weight was 20% lower. The excess calories were lost as heat rather than being stored as fat.” Regulation of fat saturation seems very robust up to a level of around 6% of calories from linoleic acid. Americans had relatively saturated body fat in 1962. By 1991, dietary and stored PUFA levels had risen substantially, and the desaturase index (DI) had greatly increased in parallel. By 1991, LA consumption had dys-regulated American SCD1 levels.
- Bear Hunting Season; Native Americans Hunted Bear When The PUFA Content Was Lowest: Bear fat was a Native American staple. Instead of hunting bear in the fall when the bear were the fattest, they waited until late winter to hunt bear – when the PUFA content of the fat was the lowest. A winter staple food was roasted venison dipped into a sauce of bear fat and maple sugar mixed so that it nearly as sweet as pure maple sugar. There is no evidence that Native Americans got fat from this , unlike those eating pure maple sugar.
- Fat Newborns; Sloth and Gluttony Part 1. The desaturase index of mother’s blood is strongly associated with newborn BMI and up until 2 years of age. This is independent from the blood sugar or insulin levels of the mother. Since these are newborns we know they didn’t become obese by overeating or couch-potatoism.
- Good PPAR, Bad PPAR. It can be clearly demonstrated in mice that feeding them a diet of 10% linoleic acid dys-regulates their fat metabolism, increasing PPAR gamma and decreasing PPAR alpha. PPAR gamma upregulates SCD1 and Elovl3. The end products of SCD1 and Elovl3 upregulate PPAR gamma. Positive feedback loop again. This mouse study is an exact parallel to what happened to humans in America between 1962 and 1991 as chronicled in The Body Fat Of Starch Eaters. PPAR gamma makes humans fat and PPAR alpha causes weight loss in humans.
- Very Long Chain Saturated and Monounsaturated Fats are another indicator that your SCD1-PPAR gamma-Elovl axis is dysregulated. High levels of them are associated with increasing severity of diabetic retinopathy, Major Depressive Disorder and Psychosis.
Hibernation
The bear is an interesting model – a large mammal, monogastric omnivore who enjoys salmon and berries. Again, sound like anyone you know?

We’ve already seen that the PUFA content of bear fat peaks in the fall, before hibernation. Is this coincidental to the availability of acorns – a source of LA – or is it actually functional. This review1 on the use of polyunsaturated fat by hibernators is quite clear that polyunsaturated fat enhances torpor:
Heterothermic mammals increase the proportion of polyunsaturated fatty acids (PUFA) in their body fats
Daniel Munro, Donald W. Thomas
prior to entering torpor. … Low-PUFA diets consistently increase the lower setpoint for body temperature and minimum metabolic rate for both hibernators and daily heterotherms. Above the lower setpoint, low-PUFA diets usually increase body
temperature and metabolic rate and decrease the duration of torpor bouts and this effect is similar for hibernators and
daily heterotherms.
In hibernators, just like starch eating Bolivian humans, a low-PUFA diet increases body temperature and metabolic rate.
The other thing that can decrease metabolic rate is the over-expression of SCD1, which turns saturated fat into unsaturated fat. Do hibernators make more SCD1 during torpor? This study2 shows that bears have a ratio of monounsaturated fat to saturated fat (a less specific type of DI than comparing oleic acid to stearic acid) of 1.09 in June and 1.55 in February, highly suggestive of an increase in SCD1 during torpor in the hibernator most like us.
To get into torpor, bears increase the amount of PUFA in their stored bodyfat and upregulate SCD1, just like Americans did between 1962 and 1991.
Hamsters!
It may not be shocking that hamsters are easier to study in a lab than bears. This 2019 paper3 does a beautiful job of detailing the metabolic changes that take place in the Syrian hamster as it goes through hibernation. The stimulus for the hamster to hibernate is a reduction in temperature. They make it cold. The hamsters go through 12-15 weeks of metabolic changes before going into deep torpor. These hamsters hibernate with food. Every five days or so, they increase their body temperature, wake up and eat a little, then they drop back into deep torpor. This is a graph of their body temperature.
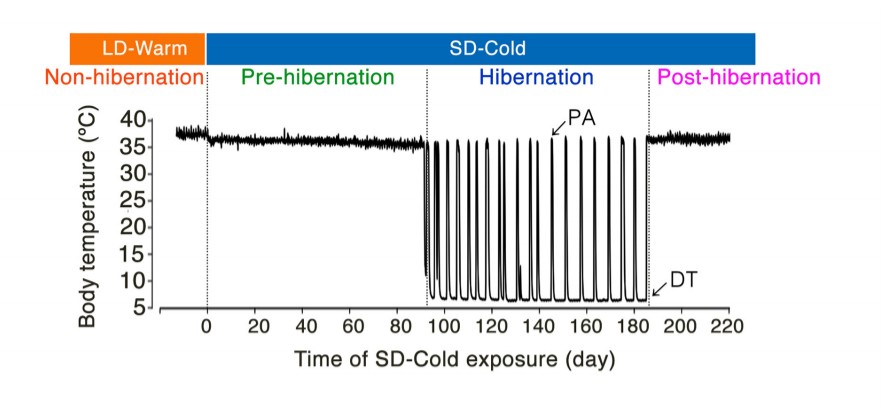
This model is a beautiful narrative framework for most of the topics covered in this thread: PPAR gamma and alpha, SCD1, UCP1 and therefore mitochondrial uncoupling and metabolic rate, and how all of those things relate to body fat levels. I’ll explain it in narrative fashion, then I’ll show you the graphs, then we’ll go through it again.
First some definitions: iWAT is inguinal white adipose tissue, a type of abdominal fat. Ppara is PPAR alpha. Ppargc1a and Ppargc1b are PPAR gamma co-regulators – a rough indicator of overall PPAR signalling. Non-HIB is non hibernating, SD-cold is when the temperature has been lowered and the animals are getting ready to hibernate, HIB-PA is the portion of hibernating while the animals body temperatures are higher and they’re awake, HIB-DT is deep torpor – low body temperature hibernation – and Post-HIB is post hibernation.
When the hamster is in non hibernating mode, PPAR alpha is low as well as PPAR activity in general. SCD1 levels are very low, but UCP1 levels are also very low due to lack of PPAR signalling. The hamster has a relatively high amount of bodyfat.
When the temperature drops, PPAR activity increases a little (light blue on the heat map is more activity than dark blue). SCD1 activity is still low, so the increase in PPAR leads to a large rise in UCP-1. The body temperature drops immediately – hamsters have a high surface to volume ratio and are affected dramatically by heat changes – but then stays stable until around the 40 day mark. During this time period – high UCP-1, low SCD1, moderate PPAR, stable body temperature – the hamsters lose significant body fat.
At the 40 day mark, the body temperature of the hamsters starts to drop steadily. SCD1 increases, UCP1 drops to almost zero and the fat mass of the hamsters is totally replenished by the twelth week. So with high SCD1 and low UCP-1 we have fat gain.
The animals go into deep torpor. SCD1 is at its highest point, PPAR alpha is a low percentage of overall PPAR signalling, UCP-1 is low and the body temperature and metabolic rate of the hamsters plummet.
Now it is time to wake up and eat. SCD1 levels drop a little and PPAR alpha levels are raised to the highest. UCP-1 levels go up. Body temperature rises. Remember than when SCD1 levels were very low a small increase in PPAR alpha lead to a large increase in UCP-1? Now that SCD1 levels are quite high, a large increase in PPAR alpha leads to a small increase in UCP-1.

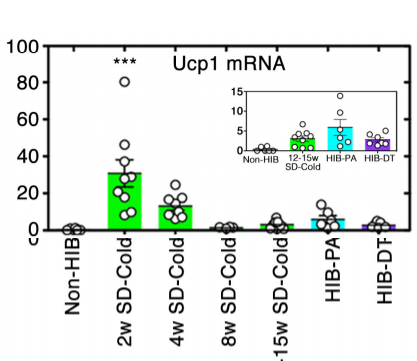
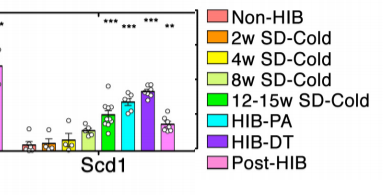
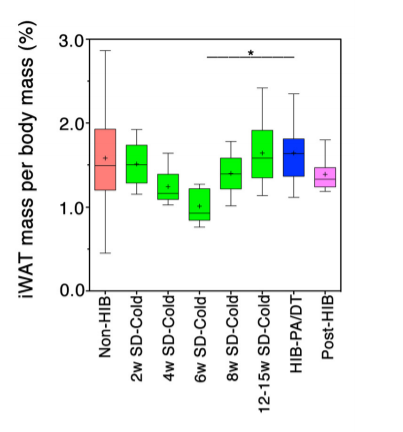
To recap:
- When SCD1 is low a small increase in PPAR signaling creates a large rise in UCP-1 and metabolic rate
- Body fat decreases when UCP-1 levels are high.
- Body fat levels rise in parallel with increasing SCD1
- When SCD1 levels are high, a large increase in PPAR alpha only leads to a small increase in UCP-1
SCD1 Is Increased by PPAR Gamma and Delta but not Alpha
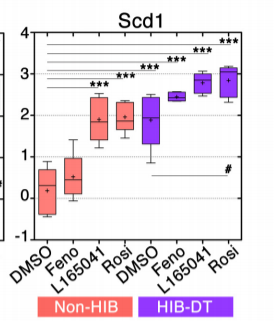
Fat cells were removed from the hamsters and cultured with either DMSO – a negative control, fenofibrate – a PPAR specific agonist (it turns it ON), L-165041 – a PPAR delta specific agonist or rosiglitazone – a PPAR gamma specific agonist. When the hamsters are in non-hibernation, activating PPAR gamma or PPAR delta substantially increase SCD1. Activating PPAR alpha does not increase SCD1. When the hamsters are in hibernation mode, SCD1 is high no matter what!
Conclusion
Deep torpor is a metabolic state that is widely distributed through the mammalian family. It is enhanced by the consumption and storage of polyunsaturated fats, in particular linoleic acid. Molecular changes that occur during deep torpor include increased expression of SCD1 and high activity of the PPAR transcription factors with a relatively low contribution from PPAR alpha. Metabolic changes during deep torpor include low body temperature, low metabolic rate and the ability to maintain body fat levels with relatively few calories.
American consumption of linoleic acid increased dramatically between 1962 and 1991. Our stored levels of linoleic acid increased, as did our desaturase index, indicating up-regulated levels of SCD1. Recent blood tests from myself and many of you show high levels of very long chain saturated and mono-unsaturated fats, suggesting that we have very high levels of PPAR gamma. The body temperature of Americans has dropped over the last 150 years. We have become very good at maintaining our fat mass.
Are you in deep torpor?
- 1.Munro D, Thomas DW. The role of polyunsaturated fatty acids in the expression of torpor by mammals: a review. Zoology. Published online March 2004:29-48. doi:10.1016/j.zool.2003.12.001
- 2.Giroud S, Chery I, Bertile F, et al. Lipidomics Reveals Seasonal Shifts in a Large-Bodied Hibernator, the Brown Bear. Front Physiol. Published online April 12, 2019. doi:10.3389/fphys.2019.00389
- 3.Chayama Y, Ando L, Sato Y, et al. Molecular Basis of White Adipose Tissue Remodeling That Precedes and Coincides With Hibernation in the Syrian Hamster, a Food-Storing Hibernator. Front Physiol. Published online January 28, 2019. doi:10.3389/fphys.2018.01973

Hi Brad,
Thanks for the great work. I changed my diet according to your recommendations about 6 month ago. Satiety is much better. My hands and feet are a lot warmer. However, my weight didn’t change at all. Still about 30 pounds too much.
Measured my DI: Came back at 2.4… So now I’m trying some of your recommendations to lower SCD1.
One simple question though: Why do you use sterculia oil? If I understand your articles correctly, you could achieve the same results using Berberine, fish oil and CLA. All of those substances are easy to buy and quite cheap.
What makes you think sterculia oil is worth the extra effort?
BR
I think that sterculia oil is another tool in the fight. It seems to be safe, and I suspect that the ability to block enzymatic activity will have different behavior than using something like berberine.
I guess I am concerned that in someone who’s level are totally dysregulated, something like berberine may not bring it under control – it may not be able to overcome the buildup of PUFA, PPAR gamma signalling, Elovl3, etc. Whereas the SO, which directly blocks the enzymatic is a surefire way to control it. My hope is that if we can get our metabolisms back on the right track, we will be able to get by without the supplements but we’ll see.
Brad
Hi
I am wondering how much sterculia do you take at a time & how much in a day??
Cherie
I am still trialing it. Right now I’m taking about 1200mg, which is a quarter teaspoon. I just tsarted taking it with Berberine – the sterculia oil seems to have slowed down my PPAR gamma signalling but I still need more PPAR alpha to get my metabolic rate up!
Brad
https://www.fightaging.org/archives/2021/03/a-popular-science-view-of-mitochondrial-uncoupling/
I don’t see a contact form. Feel free to delete this. Just wanted to send it your way.
Bear with me, let’s talk about bears (ref 2).
They are Scandinavian brown bears, and I was wondering how they get PUFAs in the Autumn… no acorns and less fish than in America. They are berry binging 50-80% of their autumn diet, negligible amount of vegetation, until lethargy and heavy weight makes them stumble over some random carcass, I guess? So, they must rely on the SCD1 for MUFA conversion?
The mentioned MUFA/SFA ratios of WAT in summer and winter were 1.09 and 1.55. Of course, the summer fat amount basis is at the minimum, and there is still huge amount of fat left midway in hibernation. Interestingly, the MUFAs are retained in tissues and WAT and short chain fatty acids left to plasma for oxidation(?). Less ROS, less signaling, less oxidative stress this way?
About execution of taking biopsy of 60kg summer bear with one word: WAU. Darting from helicopter to begin with, they have GPS on their necks.
The “day” of the bear looks interesting. Well long slept with overnight fast, breakfast only fat-protein combo, with lunch some fish and berries for dessert, the miracle of carbs begins, and dinner is feasting with anything you can find before getting to sleep with falling body temperature? Just to wake up with same muscles but >50% less fat. You can catch anything. The year of a bear… way to go.
JR
It’s unclear to me that dietary PUFA is necessary to get into torpor – increased SCD1 expression may be sufficient but the effect is certainly enhanced by linoleic acid.
Brad
Hey Brad! Thanks for sharing your ongoing research and experiments with us. I’ve been enjoying reading through and trying to get my head around things.
Are you familiar with Paul and Sou-Shing Jaminet’s “Perfect Health Diet?” The more I read of your material, the more it is seeming (to me) to fill in the rationale for some aspects of that diet approach: i.e. the emphasis on saturated fat, the “safe starches” concept, and eating them together.
I’ve been poring back through the PHD blog to see where the threads of research may have intersected over the years.
JonMarc
I’ve been to their site a couple of times, but I haven’t had time to really dig in. It seems like we are on the same wavelength, as I am generally in line with many of the thoughts of Ray Peat, although I certainly don’t agree with him on everything! I also haven’t had time to look in depth at his research and opinions.
Brad
Brad, thoughts on Protein? I’d love to have you add that to the mix.
When do you begin your Patreon!!?
Howdy Brad. I recently placed an order with you and sent you an email regarding it (I just replied to the email I received as a receipt). Could you kindly get back to me when you get the chance?
I need help/ideas!
Been in the program for several weeks, & I’ve lost about seven lbs without cravings or late night visitations from the Devil. This is a FIRST for me.
However…
I’m quite the foodie & home cook, but I have discovered that I do NOT like ghee, the butteroil, or the stearic acid powder! The taste, aroma, mouthfeel all are very off-putting to me. I ruined some fine butter, mixing it with stearic acid. I’ve mostly just choked the stearic acid down in water. But it doesn’t mix! When I tried hot water, it crystallized.
What to do??
Any amazing recipes or ideas? I hate the stuff, but I’m willing to keep trying.
Hi!
It’s admittedly a weird texture/flavor. This is why I use it with starch. I don’t know if that’s something you’re in to. For instance, I fry tortilla chips with it.
I use the butteroil to make things such as rich brioche: https://www.foodandwine.com/recipes/extra-rich-brioche
Substitute 8 Oz of the butteroil for the 2.5 sticks of butter in that recipe. I roll it into balls and freeze them when it’s done – makes a bunch of meals and I roll the doughs out into like pizza crusts or roll them into ham-n-cheese spirals.
Also, I have some new products I’m working on that will be more natural sources of stearic acid. One is rendered beef suet, which is very high but can have a bit of a strong beefy flavor. The other is fractionated butter, which isn’t AS high in stearic acid as the SA-enhanced butteroil, but it’s a lot higher than regular butter and is an all natural product, used in European layer pastries.
Brad
I had the same problem. Turned out i love cocoa butter. I fry everything in it now. Noodles, rice, potatoes, eggs steak… whatever. I even like it better than regular butter now – and it’s about 33% stearic acid.
I have thought about it this way: the mouthfeel is part of the effect on satiety. My protocol is to eat about 500g of low fat grass fed ground beef on weekdays. I add about a tablespoon of stearic acid to that, and that 500g of ground beef becomes very hard to eat, partly because of the waxy mouthfeel. Once I get that down, I do not want to think about food for next good 12 hours, and then I am already sleeping. I am not usually hungry for many hours after I wake up, so that leaves only few hours of slight hunger before my next meal. If I do not add stearic acid to my beef, I can easily eat more in one sitting, and would be hungry again after about 6 hours. I load up some surplus calories on weekends in order to prevent slowing down my metabolism, and try to avoid PUFAs as much as possible. Losing fat has never been this painless. And like the theory predicts, I seem to loose fat surprisingly fast around the midsection, even though I have very little visceral fat.
Great website and blog, thanks 👍🏻
There is so much material to digest. Can you summarise for me in a sentence the answer to:
If / when someone stops eating PUFAs, but then eats fairly ‘normally’; will the body make any use of the stored PUFA fat? How long does it take for all the PUFA to leave the body?
Thanks
This is a commonly asked question. Unfortunately I don’t have a great answer. Tucker Goodrich on Twitter has suggested over two years I think. It probably also depends on your fat mass.
Thank you!
Brad
Thanks Brad 👍🏻
I’m amazed not many people seem to be aware that PUFAs switch off ‘Fat Burning’ for a while, this is why people are hungry after eating, can’t use their stored fat, and then eat more… repeated endlessly ?
Yeah, I think that’s it!
Brad
Thanks Brad 👍🏻
I am a bit surprised that not many commentators mention the way PUFAs switch off fat burning for a little while!
I mean if you fast 3 to 4 days, eat, then another 3 to 4 days so on and so forth you’ll loose tons of fat. I’ve went from 17% bf to 11% like that in about a month. It can’t possibly take two years if you do that. As you can see your bf go down a lot. That is your fat leaving your body literally.
If you watch naked and afraid you can see fasting in action for 21 days or more. They lose tons of fat. They usually only eat protein because that’s all there is out there. It’s not rocket science if you really want to fix your body you gotta do what you gotta do. And not eat. And eat as if you were in naked and afraid. Like in the wild. Like lions. Look up the Snake Diet for some motivation into fasting. Tons of examples.
Fasting your way down to a healthy level can reset a lot of health issues people have. Trying to fix your metabolism by eating differently is kind of hard in a way. Trying to fix or heal your body earint croissants is just plain silly. Not that people are here for that. I hope not. Its ok as a diet alternarive and a great example that carbs and sat fat are not evil. But if you are here to fix your health and you body through that, why not just not eat? Give your body a break to repair and fix what you’ve done to it. And when you resume eating you’ll be all the wiser and hopefully won’t do it again and apply some of the concepts from this blog. Not eating LA over 6% etc.
I didn’t mean to write that much but I have a feeling a lot of people are here to fix there body or health and really it’s not that hard, just don’t eat, let your body heal, and then start eating healthy like your supposed to.
I hurt my small intestine by experimenting with fasting for a week a month a couple of years back. I think it really depends on other underlying conditions – you can’t blanket recommend things.
I fasted back in November. It was for 5 days. I lasted three. It was terrible. Then I got sick a couple of days after the fast.
I’ll never do it again.
I mean…. I’ll just say that
Some people need to ease into it. One day at a time. Then 2. Then 3. With eating in between. And aclimatize or get used to it over weeks or months. I did that over weeks. You shouldn’t have tried to fast for 5 days when you’ve never done it before and then feel terrible and never do it again… you shouldn’t rule it out yet that just goes against the scientific method. You should try again and again slowly and ease into it. Then after you’ve tried and failed many times and see no results then you can be confident that it didn’t work.
As Cole from the snake diet would say feeling sick after is just a sign your not healthy. Perhaps healing. All I know is I feel great after fasting. But you gotta train like anything else. Train yourself to not eat. It takes discipline.
Also your body shouldn’t get sick after fasting. It should be strong and have energy from fat to be able to continue to hunt and get food… doesn’t that make sense? How else would we still be around as a human race if our bodies were not that resilient.
If I were you I’d look at is as a challenge that your body couldn’t handle not eating for 3 days and see why that is. Was it just once? Maybe its just a matter of getting used to it. If you never try it again you might miss out on the best thing you could have done for yourself. Try it again and see if things improve… you need to try several times before you can say ok this doesn’t work.
Also drinking snake juice or electrolytes makes a huge difference, I’d try again with that and see if it’s better for you
This is it, I believe. I love all this data… Brad is no doubt brilliant but I did it all…ALL, with and without the starch and gained 10 lbs on top of the 70 I have to lose. Strangely enough my blood test showed my saturated quotient in the “good” range. I’m going back to just not eating. Short and long fasts. It’s the only thing that works for me.
Hi Brian. I know you said you tried it all, did you try berberine?
Brad
Hi Max, I’ve done a fair bit of fasting and I agree with you it’s a good way to lose fat. I suppose the issue is what is the make up of fat in your cells after the fasting and how it affects your metabolism? Be interesting to see what your Desaturase Index (DI) is now after reducing your body fat. Have you taken that blood test?
I think Brad mentioned somewhere that fasting would upregulate SCD1. Lets see if I can find it…. yes:
See the comments: https://fireinabottle.net/the-scd1-theory-of-obesity-part-2-the-post-obese-metabolism-and-what-you-can-do-about-it/
“The problem with this theory is that fasting increases SCD1. A better option might be to consume a high fat saturated fat diet (or even a pure starch/fructose/ethanol diet) with proper inhibitors and/or down-regulators in place to steadily dilute out the stored unsaturates. Time will tell the better option, I suppose.”
Brad, it would be interesting read more about this! What is the mechanism behind? We have learned that insulin upregulates SCD1, and insulin is low when fasting. It must be something else?
I’m not sure of the mechanism, but SCD1 is very dynamically regulated. I think I was referring to this paper: https://reader.elsevier.com/reader/sd/pii/S0021925819501077?token=9B44496578FBF1A29AA5B5C3FA9C4A8D6C82414E1B6E957F1AFAB9B49B71EB987EC8F24F81B86EF77BE178D2ADA4A94A
So the SCD1 is low during a fast, but it can go really high upon re-feeding (this is likely a signal to store energy coming out of lean times), especially if what you reffed on is a fat-free, high sugar diet. That upregulated SCD1 by a whopping 45 fold.
I would like to have seen the effects of refeeding a fat-free high sugar diet compared to the effects of OTHER diets coming out of a fats, but it’s unfortunately not done.
Brad
I would love to know too. I’m pretty healthy and can fast tops for 3 days before I start day dreaming of food. I haven’t fasted in a while since I reached 11% and then went up to 13-15%. Hard to tell if one is not caliper testing.
But after fasting I still was prone to getting belly fat quite easily. I think after stopping or being aware of omega 6 seed oils and trying to cut everywhere I can possibly my belly fat has quite a bit gone down. Like that fat pad that is stubborn? At one point it was over a few weeks that it was almost gone. I had a good streak of eating hardly any 6s.
I now eat more pork and chicken because my wife likes it so it’s hard to be saying no to it. Plus she likes cooking with sesame oil and doesn’t know what I know from reading this blog. But she’s coming around slowly as I insert bits of knowledge here and there. But since I’ve been eating more of pork and chicken and less watchful the pad surely has creeped back. But not as much as it once was.
Now I am trying brads supplement cocktail of berberine, cla, jugula extract?(typing off the top of my head), fish oil to see if that does something. I’m on my 3rd or 4th week. So far stopped going to the bathroom at night. Temp is around 96.8 hasn’t changed but is around 95 in the mornings not sure why. Feeling less hungry and dropped 1kg probably eating because of less hunger. My thighs have a sort of weird cramp feeling as if the muscle is eating itself. Feel like I might be loosing muscle. But my strength is up in my handstand pushups and been going up so not sure and still building muscle and strength so maybe because I haven’t been moving much and sitting down a lot due to covid not using my legs as much. Cant say thighs are visibly skinnier either.
Have had good mental clarity and positivity as well. No lack of energy but sometimes get hunger pangs. But net calories have come down. I dont need to eat as much to feel satiated. I never was a big eater anyway. But today I barely ate and was even concerned if I should stop taking it but I will probably stick to it until I am done with the pills I bought.
My main goal was to lose more fat pad but it seems its heavily related to the 6s oils. And the berberine cocktail helps suppress appetite and regulates energy consumption or something. Anyway im no expert but thats my experience so far
In the context of ppars: What about something called Tetradecylthioacetic Acid? it looks like it activates ppars with preference for the ppar-alpha:
https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0229322
There are a lot of PPAR agonists. This one is synthetic and so I think it’d be treated like a drug. Conversely, berberine is a quite specific PPAR alpha agonist, it has been used in Chinese medicine for millennia and it has a great track record of safety and effectiveness in human trials.
Brad
There’s definitely a lot of info on the negative effects of PUFA from concentrated industrial oils. What are your thoughts on PUFA from whole foods like nuts, chicken, avocados, etc.?
From https://fireinabottle.net/the-scd1-theory-of-obesity-part-2-the-post-obese-metabolism-and-what-you-can-do-about-it/
“Strategies to fix this problem include eliminating all dietary unsaturated fats to the extent possible by replacing all sources of fat with dairy, beef suet, cocoa butter or stearic acid enhanced butteroil. This includes avoiding relatively unsaturated sources of fat such as nuts, avocados, olive oil, chicken fat and even most pork fat unless it is Low-PUFA pork. Certain polyunsaturated fats have the effect of dramatically reducing SCD, as do several herbal supplements. Sterculia oil is a natural inhibitor of SCD1 that seems to have a good safety profile and is a traditionally consumed human food.”
So dietary MUFA downregulates SCD1 and hence is OK to consume?
Olive oil should be slimming and if we consume a lot of saturated fats, then maybe SCD1 is upregulated? since body does need some MUFA in phospholipids.
So, a starchy diet with oilve oil seems to be optimal?
Where am I going wrong with this logic. Thanks
Well… Saturated Fat upregulates SCD1 a TINY bit and never to where it’d get over-expressed. And sure, if you were healthy, starch and olive oil seems to work fine, but remember that the response to olive oil – increasing PPAR alpha and decreasing SCD1 – is because olive oil isn’t really optimal, it’s too much MUFA, so we have to pull some strings to safely store out. Which healthy people are capable of doing.
Once you’re over-expressing SCD1, I thin at that point olive oil is probably more MUFA than you’d want.
Brad
Thanks, Brad, for your thoughts. Not likely to make brioche, if I’m being honest…butter croissants from the store work fine. I’ve been mixing stearic acid into some home-made sour cream, & that works well!
Also…
I’m just a regular person. Not incredibly active, not incredibly scientific. I understand the concepts & enjoy reading the stuff; I want to live my nerdy life & eat easy-to-make/get foods.
Write a cookbook! We’ll all buy it.
Well…. maybe I will someday. I hear you on easy 🙂
Great stuff as always!
So an ideal DI might be around 1.1. What’s a “bad” one? 2? 10?
What kinds of numbers do people see for never-obese vs post-obese etc?
Thanks!
Well, IDK how many people who have sent out for this test are never obese! I’m seeing a lot of numbers around 2, similar to mine. I think that’s bad.
Brad
Cool thanks. It will be fascinating to see if a return to a low DI correlates with post-post-obesity (ie go back to “normal” vs having to be super careful). I’d also love to see if those people who can “eat anything” and seemingly not gain weight also consistently have a low DI.
FWIW I plan to get the test myself soon, and hopefully repeat it ~annually.
I am so excited to hear that you are looking into other options for stearic acid. I can’t stand ghee, so the butteroil doesn’t work for me. I do have powered stearic acid, but I’m same as others, hard to work it into meals. I did try making a mousse out of it by adding to heavy whipping cream, stirring together with a little bit of salt, vanilla and allulose sweetener. It binds me up quite badly if I try to eat stearic acid that hasn’t been melted first. Is that normal? I really like the mousse, there is no weird mouthfeel at all so would love to continue using. I just don’t understand why its binding. Is the body unable to use stearic acid powder unless its been melted first?
What is fractionated butter?
Would love to hear what brand of cocoa butter others are using.
Fractionated butter is when you melt butter, then slowly cool it until some of it crystallizes and some of it is still liquid. You strain off the liquid fraction, leaving only the solids. The discarded (well, I’m sure it gets used for something) liquid phase is high in MUFA and PUFA. The solid fraction is enhanced in stearic acid. It’s around 70-80% higher is SA than normal butter.
It’s only made in Europe and it’s quite expensive, but I’m hoping to have some available soon!
Brad